Bonding
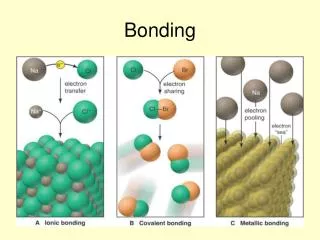
Bonding. Chemical bonding. Chemical Bonding. Graphite. Diamond. Silicon dioxide. PreDicting the type of bonding from electronegativity values. Ionic bonding typically occurs between metal and non-metal. E.g. Barium fluoride, BaF 2

Bonding
E N D
Presentation Transcript
Chemical Bonding Graphite Diamond Silicon dioxide
PreDicting the type of bonding from electronegativity values • Ionic bonding typically occurs between metal and non-metal. E.g. Barium fluoride, BaF2 • The reactivity of metals and non-metals can be assessed using electro-negativity Electro-negativity • ability of an atom in a covalent bond to attract ___________________________ to itself. shared paired of electrons
Metals generally have low electronegativity values, while non-metals have relatively high electronegativity values. • Fluorine, which has the greatest attraction for electrons in bond-forming situations (highest E value).
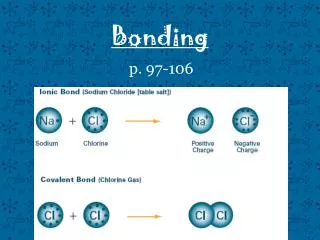
Electrons are not shared. E.g. Na+ Cl- , electron is transferred. Polar covalent bonds are covalent bonds with ionic character. Ionic bond Electrons are equally shared. E.g.Cl-Cl Non polar (pure covalent) bond Electrons are not equally shared. E.g. Atoms have different electronegativity values Polar covalent bond
Example What type of bond is the following? • N (3.0) and H (2.1) • H (2.1) and H(2.1) • Ca(1.0) and Cl(3.0) • Al (1.5) and Cl(3.0) • H (2.1) and F(4.0)
Dipole • No bond is purely ionic or covalent..they have a little bit of both characters. • When there is unequal sharing of electrons a dipole exists. Dipole - is a molecule that has 2 poles or regions with opposite charges. - is represented by a dipole arrow towards the more negative end.
Attractions Between Molecules • Besides ionic, metallic, and covalent bonds, there are also attractions between molecules • Intermolecular attractions are weaker than ionic, covalent, and metallic bonds • There are 2 main types of attractions between molecules: Van der Waals and Hydrogen
Van der Waals Forces • Van der Waals forces consists of the two weak attractions between molecules 2. dispersion forces – caused by the motion of electrons (weakest of all forces) 1. dipole interactions – polar molecules attracted to one another
Hydrogen Bond • Hydrogen Bonds are forces where a hydrogen atom is weakly attracted to an unshared electron pair of another atom
Hydrogen Bond • This other atom may be in the same molecule or in a nearby molecule, but always has to include hydrogen. INTERMOLECULAR HYDROGEN BONDING
INTRAMOLECULAR HYDROGEN BONDING • Hydrogen Bonds have about 5% of the strength of an average covalent bond • Hydrogen Bond is the strongest of all intermolecular forces
Intermolecular Attractions • A few solids that consist of molecules do not melt until the temperature reaches 1000ºC or higher called network solids (Example: diamond, silicon carbide) • A Network Solid contains atoms that are all covalently bonded to each other • Melting a network solid would require breaking bonds throughout the solid (which is difficult to do) http://library.thinkquest.org/C006669/data/Chem/bonding/inter.html
The bonding pair of electrons spends most of its time between the two atomic nuclei. • screening the positive charges from one another and enabling the nuclei to come closer together. • Negative charge on the electron pair attracts both nuclei and holds them together in a covalent bond. • When two atoms are chemically bonded, the two atoms close together have less energy and therefore are more stable than when separated. • Energy is given off by the atoms to form a bond, and energy must be supplied (absorbed) to break the bond. • A covalent bond is the result of electrostatic attraction between the nuclei of the 2 atoms and the pair of shared electrons.
H a l o g e n s F , B r , C l , I C a l c o g e n s O , S N i t r o g e n N , P C a r b o n C , S i Valence electrons and number of bonds # C o v a l e n t B o n d s* F a m i l y Number of bonds elements prefers depending on the number of valence electrons. In general - X 1 bond often 2 bond often O 3 bond often N 4 bond always C
. . Lewis Structure, Octet Rule Guidelines When compounds are formed they tend to follow the Octet Rule. Octet Rule: Atoms will share electrons (e-) until it is surrounded by eight valence electrons. Rules of the (VSEPR) game- i) O.R. works mostly for second period elements. Many exceptions especially with 3rd period elements (d-orbitals) ii) H prefers 2 e- (electron deficient) iii) :C: N: :O: :F: 4 unpaired 3unpaired 2unpaired 1unpaired up = unpaired e- 4 bonds 3 bonds 2 bonds 1 bond O=C=O NN O = O F - F iv) H & F are terminal in the structural formula (Never central) . . . . . .
N O O Cl H O O H Se O O F H C H H H O O H C C O H H O S O H H O Atomic Connectivity The atomic arrangement for a molecule is usually given. CH2ClFHNO3CH3COOH H2Se H2SO4 O3 In general when there is a single central atom in the molecule, CH2ClF, SeCl2, O3 (CO2, NH3, PO43-), the central atom is the first atom in the chemical formula. Except when the first atom in the chemical formula is Hydrogen (H) or fluorine (F). In which case the central atom is the second atom in the chemical formula. Find the central atom for the following: 1) H2O a) H b) O 2) PCl3 a) P b) Cl 3) SO3 a) S b) O 4) CO32- a) C b) O 5) BeH2 a) Be b) H 6) IO3- a) I b) O
RULES FOR DRAWING LEWIS STRUCTURES • Count the no. of valence electrons. • If the species has a –n charge, add n to the electrons • If the species has a +n charge, subtract n from the electrons • Draw a skeletal structure. • If C is present, place C at the centre. • If C is not present, place the LEAST electronegative atom at the centre. Note: H is never the in the center. • Complete the octets of the outer atoms (except for H) by adding lone pairs of electrons (including the 2 electrons shared with the central atom) • If there are any electrons left over, place them on the central atom as lone pairs. • If the central atom does not have a complete octet, rearrange lone pairs on the outer atoms to form double bonds between the central and outer atoms. Continue doing until the central atom’s octet is satisfied. • If the species is charged, place it inside brackets and write the charge outside the brackets.
Lewis structures Bond pair _____ Lone pair . . Draw Lewis structures of the following molecules: (a) H2O , NH3 , CO2 , OCl2 , PCl4+ (b) SO2 , NO+ , OCN- , COF2 , CO32- , NO2- , O3 (c) BeCl2 , BH3 , PCl5 In which of the above obey the octet rule?
Exceptions to octet rule • Molecules with an odd number of electrons • Molecules in which an atom has less than an octet • 3. Molecules in which an atom has more than an octet
1. Odd Number of Electrons NO Number of valence electrons = 11 Resonance occurs when more than one valid Lewis structure can be written for a particular molecule (i.e. rearrange electrons) NO2 Number of valence electrons = 17 Molecules and atoms which are neutral (contain no formal charge) and with an unpaired electron are called Radicals O2
2. Less than an Octet Includes Lewis acids such as halides of B, Al and compounds of Be BCl3 Group 3A atom only has six electrons around it However, Lewis acids “accept” a pair of electrons readily from Lewis bases to establish a stable octet
AlX3 Aluminium chloride is an ionic solid in which Al3+ is surrounded by six Cl-. However, it sublimes at 192°C to vapour Al2Cl6 molecules B2H6 A Lewis structure cannot be written for diborane. This is explained by a three-centre bond – single electron is delocalized over a B-H-B
Octet Rule Always Applies to the Second Period = n2 ; number of orbitals 2s, 2px, 2py, 2pz ---orbitals cannot hold more than two electrons Ne [He]; 2s2, 2px2, 2py2, 2pz2 n = 2 n = 3
Third Period ; n2 = 32 = 9 orbitals Ar [Ne]; 3s2, 3px2, 3py2, 3pz23d0 3d0 3d0 3d0 3d0 n = 3
3. More than an Octet Elements from the third Period and beyond, have ns, np and unfilled nd orbitals which can be used in bonding PCl5 P : (Ne) 3s2 3p3 3d0 Number of valence electrons = 5 + (5 x 7) = 40 10 electrons around the phosphorus SF4 S : (Ne) 3s2 3p4 3d0 Number of valence electrons = 6 + (4 x 7) = 34 The Larger the central atom, the more atoms you can bond to it – usually small atoms such as F, Cl and O allow central atoms such as P and S to expand their valency.
Multiple bonds • Bond Strength Triple bonds > Double bonds > Single bonds Page 93
The attraction between the 2 nuclei for 3 electron pairs in a triple bond is > that for 2 electron pairs in a double bond which is > than that for 1 electron pair in a single bond. (2) Triple bonds are shorter due to greater attraction between the bonding electrons and the nuclei with more electrons in the bond.
Bond strength and length of covalent bonds • Strength Triple bonds > Double bonds > Single bonds • Length Single bonds > Double bonds > Triple bonds
Coordinate (dative) bonding • In some molecules and polyatomic ions, both electrons to be shared come from the same atom forming the coordinate or dative bond. • Carbon monoxide (CO) can be viewed as containing one coordinate bond and two "normal" covalent bonds between the C atom and the O atom. How do you draw the Lewis structure? Page 94
Dissolving hydrogen chloride gas in water • Something similar happens. A hydrogen ion (H+) is transferred from the chlorine to one of the lone pairs on the oxygen atom. • The H3O+ ion is variously called the hydroxonium ion.
Other examples: • The reaction between ammonia and boron trifluoride, BF3 In BF3, there are only 6 electrons in the outer shell of boron. There is space for the B to accept a pair of electrons.
Bond Polarity • Due to difference in electronegativity value between the 2 atoms in the bond. • Unequal distribution of electron density results in small charges on the atoms ( δ+ and δ- ) Example A dipole is established when two electrical charge of opposite sign are separated by a small distance. Dipole moment
Non-polar molecule • A molecule can possess polar bonds and still non-polar. • Check the Geometry of the molecule: • The polar bonds are arranged symmetrically so as to give zero net direction of charge. i.e. Overall dipoles cancel so that there is no overall dipole.
Non-polar & polar covalent bonds Non-polar Covalent bond • No difference in electronegativity value – bond consists of 2 ____________ atoms. • _______ net charge. Examples : Polar Covalent bond • Due to the difference in electronegativity value – bond consists of 2 ____________ atoms. • _______ net charge. Examples:
polar molecule • In the water molecule, • O-H bonds are significantly polar • The bent structure makes the distribution of those polar bonds asymmetrical.
Some molecules have very low polarity - so low as to be regarded as non-polar,
For CO2 each C-O bond is polar since O is more electronegative than C. Why is the molecule non-polar?
VSEPR theory • The shapes of simple molecules and ions can be determined by using the Valence Shell Electron Repulsion (VSEPR) theory. • Electron pairs around the central atom repel each other • Bonding pairs and lone pairs arrange themselves to be as far apart as possible
Principles of VSEPR theory • Find the number of electron pairs / charge centres in the valence shell of the central atom. • Electron pairs / charge centres repel each other to the positions of minimum energy in order to gain maximum stability. • Pairs forming a double or triple bond act as a single bond • Non-bonding pairs repel more than bonding pairs.
Practice • Methane (CH4) – tetrahedral • Ammonia (NH3) – pyramidal • Water (H2O) – bent • Carbon Dioxide (CO2) - linear
Valence Shell Electron-Pair Repulsion Theory (VSEPR) Procedure • Sum the total Number of Valence Electrons Drawing the Lewis Structure 2. The atom usually written first in the chemical formula is the Central atom in the Lewis structure • Complete the octet bonded to the Central atom. However, elements in the third row have empty d-orbitals which can be used for bonding. • If there are not enough electrons to give the central atom an octet try multiple bonds. Predicting the Shape of the Molecule • Sum the Number of Electron Domains around the Central Atom in the Lewis Structure; Single = Double = Triple Bonds = Non-Bonding Lone Pair of Electrons = One Electron Domain • From the Total Number of Electron Domains, Predict the Geometry and Bond Angle(s); 2 (Linear = 180º); 3 (Trigonal Planar = 120º); 4 (Tetrahedral = 109.5º); 5 (TrigonalBipyramidal = 120º and 90º); 6 (Octahedral = 90º) • Lone Pair Electron Domains exert a greater repulsive force than Bonding Domains. Electron Domains of Multiple Bonds exert a greater repulsive force than Single Bonds. Thus they tend to compress the bond angle.
Lone pairs and bond angles Lone pairs are held closer to the nucleus than the bonding pairs. The distance between the lone pair electrons and the bonding pairs of electrons is shorter than the distance between the bonding pairs to each other. Repulsion due to lone pairs causes the bond angles to become smaller