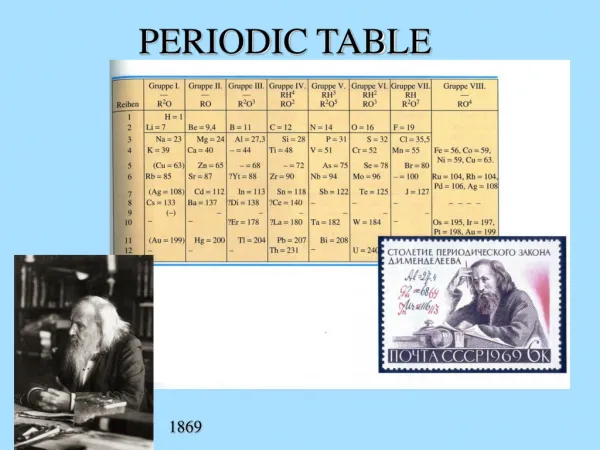
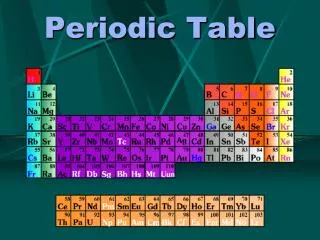
Periodic Table
Periodic Table. Electron Configurations. Aufbau Principle – electrons occupy the orbitals of lowest energy first Hund’s Rule - electrons fill orbitals in the same energy level first with electrons spinning the same direction and then the other direction

Periodic Table
E N D
Presentation Transcript
Periodic Table Electron Configurations
Aufbau Principle – electrons occupy the orbitals of lowest energy first • Hund’s Rule - electrons fill orbitals in the same energy level first with electrons spinning the same direction and then the other direction • Valence electrons are the ones in the highest occupied energy level. • N, P, As, Bi have the same outer energy level configurations.
The maximum number of electrons that can occupy one orbital is ____ • 2 • Give the symbol and name of the elements that correspond to these configurations of an atom. • 1s 22s 22p 3 ____ ___________ • N Nitrogen • 1s 22s 22p 4 ____ ___________ • O Oxygen • 1s22s22p63s23p1 ____ ___________ • Al Aluminum
1s 22s 22p 63s 23p 2 ____ ___________ • Si Silicon • 1s 22s 22p 63s 1 ____ ___________ • Na Sodium • 1s 22s 22p 63s 23p 64s 1 ____ ___________ • K Potassium • 1s 22s 22p 63s 23p 63d 24s 2 ____ ___________ • Ti Titanium
How many valence electrons do the following elements have? • oxygen ___________ • 6 • aluminum ___________ • 3 • sodium ___________ • 1 • barium ___________ • 2
The next atomic orbital in the series 1s, 2s, 2p, 3s, 3p is ____ • 4s • The electron configuration for fluorine is ________ • 1s22s22p5 • The correct electron configuration for • silicon, atomic number 14 is __________ • 1s 22s 22p 63s 23p 2 • The configuration for the outermost (valence) energy level • in Ca is _____ • 4s2
Which orbital configuration is the valence electron configuration for each of these elements? • sulfur _____ • s2p4 • germanium _____ • s2p2 • beryllium _____ • s2 • krypton _____ • s2p6 • strontium _____ • s2
Give electron configurations for atoms of these elements • Na _________________ • 1s22s22p63s1 • S _________________ • 1s22s22p63s23p4 • Mg _________________ • 1s22s22p63s2 • Ne _________________ • 1s22s22p6
K _________________ • 1s22s22p63s23p64s1 • selenium _________________ • 1s22s22p63s23p63d104s24p4 • vanadium _________________ • 1s22s22p63s23p63d34s1 • nickel _________________ • 1s22s22p63s23p63d84s2 • calcium _________________ • 1s22s22p63s23p64s2