Acid-Base Indicators
Acid-Base Indicators. Measuring pH. Now that we know how to calculate pH of a solution, how do we measure it in the lab? The following slides will provide numerous examples of ways to measure pH in the laboratory. pH testing. There are several ways to test pH Blue litmus paper (red = acid)

Acid-Base Indicators
E N D
Presentation Transcript
Measuring pH • Now that we know how to calculate pH of a solution, how do we measure it in the lab? • The following slides will provide numerous examples of ways to measure pH in the laboratory.
pH testing • There are several ways to test pH • Blue litmus paper (red = acid) • Red litmus paper (blue = basic) • pH paper (multi-colored) • pH meter (7 is neutral, <7 acid, >7 base) • Universal indicator (multi-colored) • Indicators like phenolphthalein • Natural indicators like red cabbage, radishes
Paper testing • Paper tests like litmus paper and pH paper • Put a stirring rod into the solution and stir. • Take the stirring rod out, and place a drop of the solution from the end of the stirring rod onto a piece of the paper • Read and record the color change. Note what the color indicates. • You should only use a small portion of the paper. You can use one piece of paper for several tests.
pH meter • Tests the voltage of the electrolyte • Converts the voltage to pH • Very cheap, accurate • Must be calibrated with a buffer solution
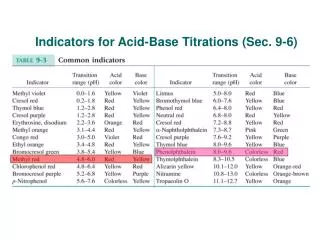
pH indicators • Indicators are dyes that can be added that will change color in the presence of an acid or base. • Most are large molecules that act as weak acids. The color changes occur when the molecules is ionized (acting as an acid) or un-ionized (acting as a base). • Some indicators only work in a specific range of pH. Choosing the right one is important! • Once the drops are added, the sample is ruined • Some dyes are natural, like radish skin or red cabbage
Choosing the Correct Indicator • Not all indicators shift colors at pH 7. • Many change colors over a wide range of pH values. • When testing a sample for acidity or basicity it is best to choose an indicator that has a color change close to pH 7 • Or use pH paper • Or use a pH meter to get a digital reading
Sample Indicator pH Ranges • Crystal Violet = yellow (pH 1) blue • Cresol Red = red (pH 2) yellow (pH 8) red • Thymol Blue = red (pH 3) yellow (pH 8) green/blue • Bromophenol blue = yellow (pH 4) blue • Methyl orange = red (pH 4.5) orange/yellow • Bromcresol green = yellow (pH 4.5) green/blue • Methyl red = pink (pH 5.5) yellow • Bromcresol purple = yellow (pH 6) purple • Alizarin = yellow (pH 6.5) red (pH 12) purple • Bromthymol blue = yellow (pH 7 ) green/blue • Phenol red = yellow (pH 7.2) red • Phenolpthalein = clear (pH 8.3) pink • Thymolphtalien = clear (pH 10) blue • Alizarin yellow GG = orange (pH 10.5) red • Universal Indicator = red (pH 4) orange (pH 6) yellow (pH 7) green (pH 8) blue (pH 9) purple