Acid Base Indicators
Experiment #6. Acid Base Indicators. What are acids and bases?. There are many different definitions for classifying a substance as an acid or a base. Definitions are based on different theories. Arrhenius theory of acids and bases. Lowry-Bronsted theory of acids and bases.

Acid Base Indicators
E N D
Presentation Transcript
Experiment #6 Acid Base Indicators
What are acids and bases? There are many different definitions for classifying a substance as an acid or a base. Definitions are based on different theories Arrhenius theory of acids and bases Lowry-Bronsted theory of acids and bases Lewis’s theory of acids and bases
Arrhenius theory of acids and bases Proposed in 1884 by Svante Arrhenius An acid is a substance that contains hydrogen and produces H+ in aqueous Solution. A base is a solution that contains the OH group and produces Hydroxide ions OH-, in aqueous solution Neutralization is defined as the combination of H+ ions with OH- ions to form H2O molecules. H+ (aq) + OH-(aq) H2O(l) Very Limited in Scope!!
Lowry-Bronsted theory Proposed in 1923 by N. Bronsted and T.M.Lowry An acid is defined as a proton donor, H+, and a base is Defined as a proton acceptor An acid-base reaction is the transfer of a proton from an acid to a base The complete ionization of hydrogen chloride, HCl in water is an acid-base reaction Step 1: HCl(aq) H+(aq) + Cl-(aq) (Arrehnius) Step 2: H+ (aq) + H2O(l) H3O+ Overall: HCl(aq) + H2O(l) H3O+(aq) + Cl-(aq) (LB)
Overall: HCl(aq) + H2O(l) H3O+(aq) + Cl-(aq) (LB) conjugate acid conjugate base acid base Overall: NH3(aq) + H2O(l) NH4+(aq) + OH-(aq) (LB) conjugate acid conjugate base base acid Whether water can act as an acid or base depends on the other species present
Autoionization of water H2O (l) + H2O(l) H3O+ (aq) + OH-(aq) Simplified notation H2O(l) H+ (aq) + OH-(aq) Water is said to be amphiprotic
Strength of acids Acids and bases are classified as strong or weak depending on the extent to which they can ionize. Higher ionization ≡ Strong Strong acids and strong bases are completely ionized Weak acids and weak bases are partially ionized Strong acid does not necessarily mean that it is the most corrosive acid. HF is a weak acid but it is one of the most corrosive acids. Strength of some acids: HClO4 > HI > HBr > HCl > HNO3 H2SO4 > HNO3
Lewis theory Proposed in 1923 by G. N. Lewis An acid is any species that can accept a share in an electron pair. A base is any species that can make available or donate a share in an electron pair. A more broader definition. There is no need for the molecule to have hydrogens.
What happens when salts are dissolved in water? HCl(aq) + NaOH(aq) NaCl(aq) + H2O(aq) Acid Base Salt Water NaCl(s) + H2O NaOH(aq) + HCl(aq) Salt Water Base Acid Depending on the strength of the acid and base that are formed in solution, the medium will be either acidic or basic
How do I know if the resulting solution is acidic or basic? Measure pH of the solution What is pH? It is the measure of hydrogen ions in solution
What is pH? pH=14.0 Basic Neutral pH=7.0 Acidic pH=1.0
What are some of the ways to measure pH? 1. Using an acid-base indicator 2. Using pH paper 3. Using pH meter 4. ……………..
What are acid-base indicators? How do they work? An organic dye, whose color depends on the concentration of H3O+ or pH HIn(aq) + H2O(l) H3O+(aq) + In-(aq) conjugate acid conjugate base acid base When the indicator is added to the solution of interest, depending on the pH of the solution, the indicator stays in either its acid form or its conjugate form.
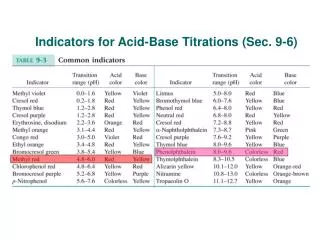
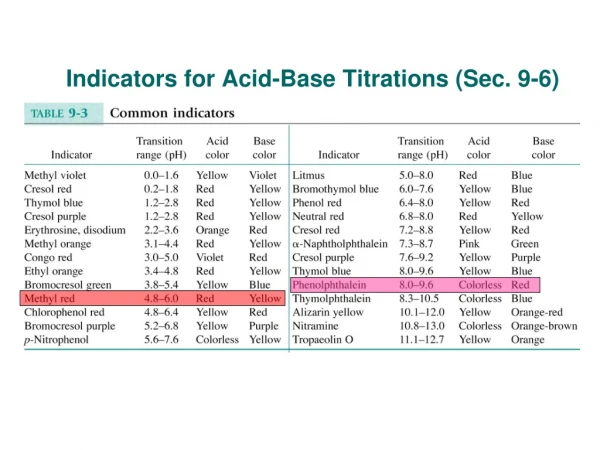
Indicator"Acidic" Color"Basic" color (MO)methyl orange Red below pH 4.0 Yellow above pH 4.4 (BG)bromcresol green Yellow below pH 4.5 Blue above pH 4.8 (BB)bromthymol blue Yellow below pH 6.9 Blue above pH 7.3 (TB)thymol blue Yellow below pH 8.0 Blue above pH 8.4 (P)Phenolphthalein Colorless below pH 9.3 Pink above pH 9.7 (AY)alizarin yellow Yellow below pH 10.8 Red above pH 11.2 Indicators cannot give a particular numerical value for pH. They can only give a range for the pH of the solution. If we are to pick a suitable indicator for following the pH change in the reaction, We have to pick one that is suitable over the pH range of the reaction.
Ionization of acids All acids undergo ionization. Strong acids ionize more than the weak ones. Strong acids are almost 100% ionized. Since the weak acids are partially ionized, the measure of the extent of ionization is given by an expression called acid ionization constant, Ka. In our experiment today, we would like to measure the Ka for a weak monoprotic acid.
What is a monoprotic acid? Usually acids have one or more hydrogen atoms (H) which they end up donating as protons (H+). # of H atoms Name Example 1 monoprotic acid (HA) HCl – hydrochloric acid CH3COOH - acetic acid 2 diprotic acid (H2A) H2SO4 – sulfuric acid 3 triprotic acid (H3A) H3PO4 – Phosphoric acid
Ionization constant of an acid For a monoprotic weak acid (HA) dissolved in water, HA(aq) + H2O(l) H3O+(aq) + A-(aq) conjugate acid conjugate base acid base
Ionization constant of an acid Ionization constant of the acid
Ionization constant of an acid Taking log of the equation on both sides,
Ionization constant of an acid Multiplying both sides of the equation by -1
Ionization constant of an acid Henderson-Hasselbach equation In the above equation, if we can make the [A-] = [HA]
Vinitial Vfinal- Vinitial =Vused Add Vused amount of water to the solution in the volumetric flask 0.2 M NaOH Vfinal Mix the solution in volumetric flask to the Solution in the erlenmeyer flask 100mL 1.0 g of HA In 100mL of water Measure the pH of the final solution with indicators and pH paper 250mL 250mL 50 mL of HA + 2 drops phenolphthalein