Exploring Periodic Trends in Atomic Properties: Setup & Analysis
60 likes | 169 Views
Understand the setup of the periodic table, trends in atomic radius, ionization energy, electron affinity, and electronegativity. Learn how these factors change across periods and groups in elements.

Exploring Periodic Trends in Atomic Properties: Setup & Analysis
E N D
Presentation Transcript
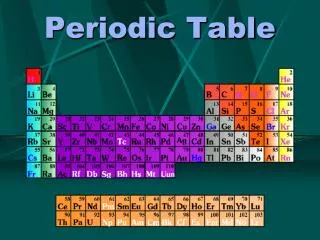
Periodic Trends Atomic Radius: 1/2 distance between 2 nuclei of the same element. Group Trend: Increases Why: e-are being added to new energy levels further away, electron shielding occurs, so the nucleus can not pull electrons in a close. Period Trend: Decreases → Why: e-are being added to the same energy level. As the nucleus becomes stronger, the electrons are drawn in closer.
Ionization Energy: The energy required to remove an e-. Group Trend: Decrease → Why: e-are further away (shielded) and easier to take away. Period Trend: Increase → Why: e-are closer to nucleus + harder to remove. Atoms are closer to having a complete valence shell and want to gain not lose e-.
Electron Affinity : ability to attract e- Group Trend: Decreases Why : Electrons are harder to bring in further away with the nucleus pulling less. Period Trend: Increases → due to a stronger nucleus Why: e- are easier to bring in Atoms want to complete their valence shell. Electronegativity: ability to attract e-in a bond → scale of O-4 F=4.0
Ion Size cations (+) : lose valence e' Effect on Radius: decrease due to loss of energy level. anions (+): gain electrons increases due to addition Effect on Radius : l , of e- to the same energy level and not changing the strength of the nucleus.