Discovering 19th Century Elements: Naming, Symbols, and Classification
This overview highlights the discovery of chemical elements in the 19th century, focusing on their origins and naming conventions. Chemists were granted the right to name new elements, with unique examples including Polonium, Uranium, and Einsteinium, each reflecting significant historical or cultural ties. The transition from names to symbols led to a systematic representation of elements, employing first letters, pairs, or Latin roots. Elements are categorized into metals, non-metals, and metalloids, with families such as alkali metals and noble gases showcasing their diverse properties.

Discovering 19th Century Elements: Naming, Symbols, and Classification
E N D
Presentation Transcript
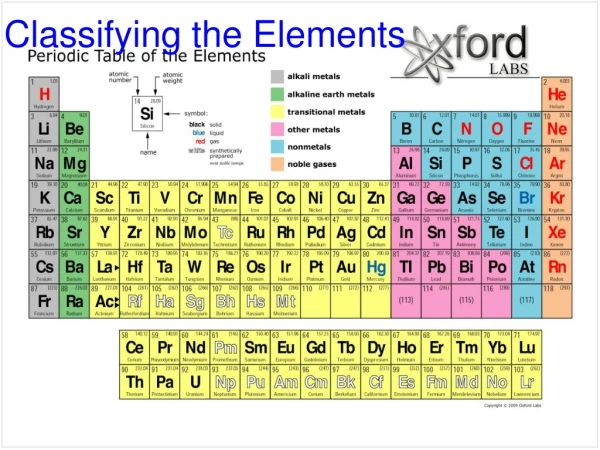
Classifying Elements Topic 4
Element Symbols • The names of elements that were discovered in the 19th century came from many different sources. • The first chemist to report a new element had the right to name it. • Polonium was named after Poland, Marie Curie’s country of birth • Uranium was named after the planet Uranus • Einsteinium was name after Albert Einstein • Instead of writing these names out, chemists developed symbols
Element Symbols • Some elements use the first letter of their name • Ex. Hydrogen - H • Some elements use the first two letters of their name • Ex. Calcium - Ca • Some elements use two letters from their name • Ex. Magnesium - Mg • Some elements use letters from their Latin name • Ex. Lead – Pb (Plumbum)
Sorting Elements • Metals • Shiny, malleable, ductile. • They conduct electricity • Non-metals • Can be a solid or a gas • Solid non-metals are dull and brittle. • Most non-metals do not conduct electricity • Metalloids • Have both metallic and non-metallic properties.
Chemical Families • Alkali Metals • Group 1 on the periodic table • So reactive that they need special storage • Only have 1 electron in their outer shell • Alkaline Earth Metals • Group 2 on the periodic table • Reactive, but not as reactive as Alkali metals • Halogens • Group 17 on the periodic table • React vigorously with other elements, so they are naturally found in compounds • Noble Gases • Group 18 on the periodic table • Very unreactive – their outer shells are full of electrons
What to do now… • Work on Lab write-up (due Monday) • Work on Trading Cards (due Monday) • Work on Chemistry Dictionary