Cone Beam Computed Tomography
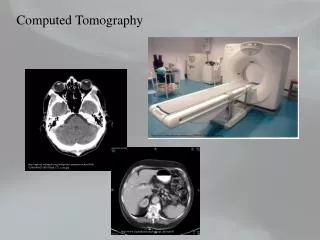
Cone Beam Computed Tomography. Cone-beam Units. NewTom 3G by AFP. Galileos by Sirona. MercuRay by Hitachi. 3D Accuitomo by J. Morita. I-CAT by ISI. Iluma by IMTEC. Comparative Dosimetry. Effective dose ( m Sv). Dental Applications. Broad acceptance in dentistry last five years

Cone Beam Computed Tomography
E N D
Presentation Transcript
Cone Beam Computed Tomography Bureau of Radiological Health
Cone-beam Units NewTom 3G by AFP Galileos by Sirona MercuRay by Hitachi 3D Accuitomo by J. Morita I-CAT by ISI Iluma by IMTEC
Comparative Dosimetry Effective dose (mSv) Source; Stuart White- UCLA School of Dentistry
Dental Applications Broad acceptance in dentistry last five years • Orthodontic treatment planning • Dental implants • Temporomandibular joints for osseous degenerative changes • Evaluation of wisdom teeth vs. mandibular nerve • Disease Source; Stuart White- UCLA School of Dentistry
Cephalometric Analysis Source; Stuart White- UCLA School of Dentistry
Bureau Issues • How to regulate these sources? • Appears that FDA has classified them as CT • If CT, subject to QA requirements • If CT, limited licensed technologists prohibited from operating unit • Your thoughts? Source; Stuart White- UCLA School of Dentistry
Suggested Regulatory Approach • Advise all CBCT registrants that FDA classifies as CT (Once we officially confirm) • Advise registrants that only the Dentist, Physician, Dental Hygienist(?) or Diagnostic Technologist can operate CBCT • Provide registrants 90 days to develop and 180 days to implement alternative QA program as permitted by N.J.A.C. 7:28-22.3(g). Source; Stuart White- UCLA School of Dentistry