Stimulated emission
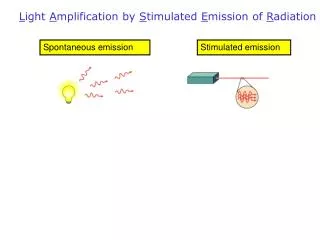
L ight A mplification by S timulated E mission of R adiation. Spontaneous emission. Stimulated emission. Lecture VIII. LASER. Energy level diagram. The possible energies which electrons in the atom can have is depicted in an energy level diagram. . T he operation of the Laser.

Stimulated emission
E N D
Presentation Transcript
Light Amplification by Stimulated Emission of Radiation Spontaneous emission Stimulated emission
Lecture VIII LASER
Energy level diagram • The possible energies which electrons in the atom can have is depicted in an energy level diagram.
The operation of the Laser • In 1958, Charles Townes and Arthur Schawlow theorized about a visible laser, an invention that would use infrared and/or visible spectrum light. • Light Amplification by Stimulated Emission of Radiation- (LASER). • Properties of Lasers • Produce monochromatic light of extremely high intensity.
The operation of the Laser (Pumping the Laser)
The operation of the Laser absorption
The operation of the Laser Spontaneous emission
The operation of the Laser Spontaneous emission • Incoherent light • Accidental direction
The operation of the Laser Stimulated emission
The operation of the Laser Light: Coherent, polarized The stimulating and emitted photons have the same: frequency phase direction
Two level system E2 E2 hn hn hn absorption Spontaneous emission Stimulated emission E1 E1 hn =E2-E1
E2 E1 Boltzmann’s equation example: T=3000 K E2-E1=2.0 eV • n1 - the number of electrons of energy E1 • n2 - the number of electrons of energy E2
Einstein’s coefficients Probability of stimulated absorption R1-2 R1-2 = r (n) B1-2 Probability of stimulated and spontaneous emission : R2-1 = r (n) B2-1 + A2-1 assumption: n1 atoms of energy e 1 and n2 atoms of energy e 2 are in thermal equilibrium at temperature T with the radiation of spectral density r (n): n1 R1-2 = n2 R2-1 n1r (n) B1-2 = n2 (r (n) B2-1 + A2-1) E2 E1
According to Boltzman statistics: • r (n) = = Planck’s law B1-2/B2-1 = 1
The probability of spontaneous emission A2-1 /the probability of stimulated emission B2-1r(n ): • Visible photons, energy: 1.6eV – 3.1eV. • kT at 300K ~ 0.025eV. • stimulated emission dominates solely when hn /kT <<1! • (for microwaves: hn <0.0015eV) • The frequency of emission acts to the absorption: • if hn /kT <<1. x~ n2/n1
E2 E1 Condition for the laser operation Ifn1 > n2 • radiation is mostly absorbed absorbowane • spontaneous radiation dominates. if n2>> n1 - population inversion • most atoms occupy level E2, weak absorption • stimulatedemission prevails • light is amplified Necessary condition:population inversion
E2 E1 How to realize the population inversion? Thermal excitation: impossible. The system has to be „pumped” Optically,electrically.
The Uncertainty Principle Measurement disturbes the system
The Uncertainty Principle • Classical physics • Measurement uncertainty is due to limitations of the measurement apparatus • There is no limit in principle to how accurate a measurement can be made • Quantum Mechanics • There is a fundamental limit to the accuracy of a measurement determined by the Heisenberg uncertainty principle • If a measurement of position is made with precision Dx and a simultaneous measurement of linear momentum is made with precision Dp, then the product of the two uncertainties can never be less than h/2p
The Uncertainty Principle Virtual particles: created due to the UP
The laser operation Three level laser E3 Fast transition E2 Laser action E1 • 13 pumping • spontaneous emission 3 2. • state 2 is a metastable state • population inversion between states 2 and 1. • stimulated emission between 2 i 1.
E3 szybkie przejścia The laser operation E2 akcja laserowa E1 - optical pumping - occupation of E3 of a short life time, 10-8s. It is a band, the metastable and ground states are narrow : - electrons are collected on E2: population inversion - stimulated emission (one photon emitted spontaneously starts the stimulated radiation ) - Beam of photons moves normally to the mirrors – standing wave.
ruby laser • discovered in 60-ies of the XX century. • ruby (Al2O3) monocrystal, Crdoped.
4T1 2T2 Energy 4T2 2E LASING 4A2 Ruby laser • Akcja laserowa z jonów Cr3+, zawartych w rubinie . • Laser trzypoziomowy. Al2O3 • optical pumping: 510-600nm and 360-450nm. • fast transition on 2E. • lasing: 2E on4A2, • 694nm Cr+ rapid decay
Ruby laser First laser: Ted Maiman Hughes Research Labs 1960