Chronic Effects of PCP Treatment on Prepulse Inhibition in Rats
E N D
Presentation Transcript
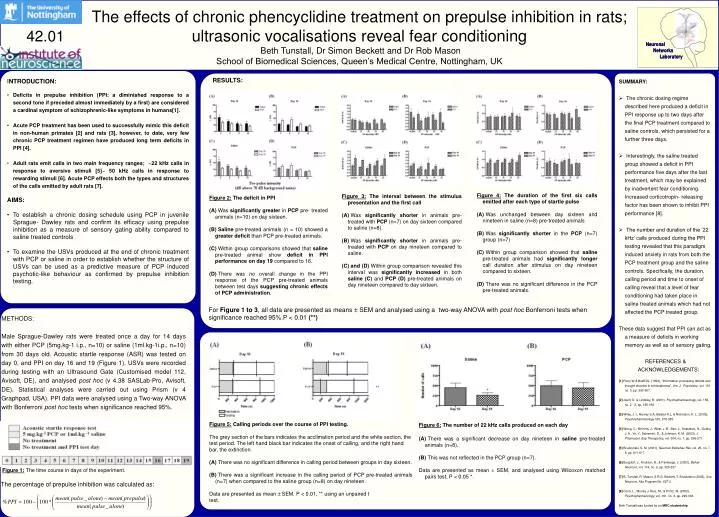
Figure 4:The duration of the first six calls emitted after each type of startle pulse • Was unchanged between day sixteen and nineteen in saline (n=8) pre-treated animals • Was significantly shorter in the PCP (n=7) group (n=7) • Within group comparison showed that saline pre-treated animals had significantly longer call duration after stimulus on day nineteen compared to sixteen. • There was no significant difference in the PCP pre-treated animals. • Figure 3: The interval between the stimulus presentation and the first call • Was significantly shorter in animals pre-treated with PCP (n=7) on day sixteen compared to saline (n=8). • Was significantly shorter in animals pre-treated with PCP on day nineteen compared to saline. • and (D) Within group comparison revealed this interval was significantly increased in both saline (C) and PCP(D) pre-treated animals on day nineteen compared to day sixteen. • Figure 2: The deficit in PPI • Was significantly greater in PCP pre- treated animals (n=10) on day sixteen. • Saline pre-treated animals (n = 10) showed a greater deficit than PCP pre-treated animals. • Within group comparisons showed that saline pre-treated animal show deficit in PPI performance on day 19 compared to 16. • There was no overall change in the PPI response of the PCP pre-treated animals between test days suggesting chronic effects of PCP administration. For Figure 1 to 3, all data are presented as means ± SEM and analysed using a two-way ANOVA with post hoc Bonferroni tests when significance reached 95%.P < 0.01 (**) • Figure 5: Calling periods over the course of PPI testing. • The grey section of the bars indicates the acclimation period and the white section, the • test period. The left hand black bar indicates the onset of calling, and the right hand • bar, the extinction. • There was no significant difference in calling period between groups in day sixteen. • There was a significant increase in the calling period of PCP pre-treated animals (n=7) when compared to the saline group (n=8) on day nineteen. • Data are presented as mean ± SEM. P < 0.01, ** using an unpaired t • test. • Figure 6:The number of 22 kHz calls produced on each day • There was a significant decreaseon day nineteen in saline pre-treated animals (n=8). • This was not reflected inthe PCP group (n=7). • Data are presented as mean ± SEM, and analysed using Wilcoxon matched pairs test, P < 0.05 *. Figure 1: The time course in days of the experiment. The percentage of prepulse inhibition was calculated as: The effects of chronic phencyclidine treatment on prepulse inhibition in rats; ultrasonic vocalisations reveal fear conditioning Beth Tunstall, Dr Simon Beckett and Dr Rob Mason School of Biomedical Sciences, Queen’s Medical Centre, Nottingham, UK 42.01 RESULTS: • INTRODUCTION: • Deficits in prepulse inhibition (PPI: a diminished response to a second tone if preceded almost immediately by a first) are considered a cardinal symptom of schizophrenic-like symptoms in humans[1]. • Acute PCP treatment has been used to successfully mimic this deficit in non-human primates [2] and rats [3], however, to date, very few chronic PCP treatment regimen have produced long term deficits in PPI [4]. • Adult rats emit calls in two main frequency ranges; ~22 kHz calls in response to aversive stimuli [5]~ 50 kHz calls in response to rewarding stimuli [6]. Acute PCP effects both the types and structures of the calls emitted by adult rats [7]. • AIMS: • To establish a chronic dosing schedule using PCP in juvenile Sprague- Dawley rats and confirm its efficacy using prepulse inhibition as a measure of sensory gating ability compared to saline treated controls • To examine the USVs produced at the end of chronic treatment with PCP or saline in order to establish whether the structure of USVs can be used as a predictive measure of PCP induced psychotic-like behaviour as confirmed by prepulse inhibition testing. • SUMMARY: • The chronic dosing regime described here produced a deficit in PPI response up to two days after the final PCP treatment compared to saline controls, which persisted for a further three days. • Interestingly, the saline treated group showed a deficit in PPI performance five days after the last treatment, which may be explained by inadvertent fear conditioning. Increased corticotropin- releasing factor has been shown to inhibit PPI performance [8]. • The number and duration of the ’22 kHz’ calls produced during the PPI testing revealed that this paradigm induced anxiety in rats from both the PCP treatment group and the saline controls. Specifically, the duration, calling period and time to onset of calling reveal that a level of fear conditioning had taken place in saline treated animals which had not affected the PCP treated group. • These data suggest that PPI can act as a measure of deficits in working memory as well as of sensory gating. • REFERENCES & ACKNOWLEDGEMENTS: • [1] Perry W & Braff DL. (1994), “Information-processing deficits and thought disorder in schizophrenia”, Am. J. Psychiatry., vol. 151, no. 3, pp. 363-367. • [2] Javitt, D. & Lindsley, R. (2001), Psychopharmacology, vol. 156, no. 2 - 3, pp. 165-168. • [3] Wiley, J. I., Harvey S.A, Balster R.L, & Nicholson, K. L. (2003), Psychopharmacology 165, 378-385. • [4] Wang, C., McInnis, J., West, J. B., Bao, J., Anastasio, N., Guidry, J. A., Ye, Y., Salvemini, D., & Johnson, K. M. (2003), J. Pharmacol. Exp Theraputics, vol. 304, no. 1, pp. 266-271. • [5] Brudzynski, S. M. (2001), Neurosci Biobehav Rev. vol. 25, no. 7-8, pp. 611-617. • [6] Burgdorf, J., Knutson, B., & Panksepp, J. (2000), Behav Neurosci, vol. 114, no. 2, pp. 320-327. • [7] B. Tunstall, R. Mason, S.R.G. Beckett, T. Shackleton (2003), Soc. Neurosci. Abs Program No. 627.3. • [8] Conti, L., Murray J, Ruiz, M., & Printz, M. (2002), Psychopharmacology, vol. 161, no. 3, pp. 296-303. • Beth Tunstall was funded by an MRC studentship. METHODS: Male Sprague-Dawley rats were treated once a day for 14 days with either PCP (5mg.kg-1 i.p., n=10) or saline (1ml.kg-1i.p., n=10) from 30 days old. Acoustic startle response (ASR) was tested on day 0, and PPI on day 16 and 19 (Figure 1). USVs were recorded during testing with an Ultrasound Gate (Customised model 112, Avisoft, DE), and analysed post hoc (v 4.38 SASLab-Pro, Avisoft, DE). Statistical analyses were carried out using Prism (v 4 Graphpad, USA). PPI data were analysed using a Two-way ANOVA with Bonferroni post hoc tests when significance reached 95%.