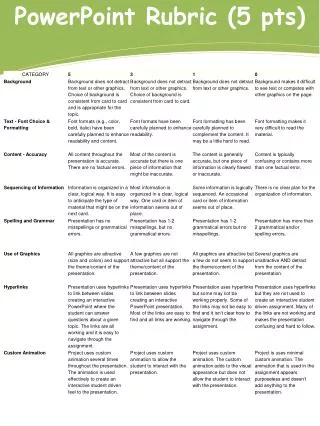
PowerPoint Rubric
PowerPoint Rubric. Bys: Emily Gregg. Democritus <3 .

PowerPoint Rubric
E N D
Presentation Transcript
PowerPoint Rubric Bys: Emily Gregg
Democritus <3 • Where scientific discoverys are concerned one can always expect new ideas and concepts to come up. There might be one scientist who’s given an explanation about how a certain thing works. The same topic might be contradicted by another scientist or a person knowledgeable about the topic, claiming something that is completely different or opposite to the original idea. One such topic is the Democritus Atomic Theory. This was a theory that was arrived at by Democritus, who was Greek by origin. Democritus was a famous personality in Greece because of his many philosophical ideologies. He was regarded with respect and was a powerful figure in his days. His theories about atoms are still studied today, although there have been many people who have claimed that his theory does not hold the actual truth.
Democritus <3 • The democritus atomic theory revolves around the atoms that are present in the atmosphere. These are about the atoms that are present in all the forms of existence for instance, solid or liquid. This theory states that these atoms are all individually created and cannot be separated, no matter what scientific procedures are applied. The Democritus Atomic Theory is pretty much clear and the ideas are portrayed in manner that can be easily understood. The theory also claims that there is no specific size that can be attributed to the various atoms and that they can differ depending on the various factors influencing them. It can be arrived at that the atoms cannot be split no matter what their size. So we see how the Democritus Atomic theory explains the existence of atoms and also its features
John Dalton • John Dalton (1766–1844) was born into a modest quaker family in cumberland, england, and earned his living for most of his life as a teacher and public lectur, beginning in his village school at the age of 12. After teaching 10 years at a quaker boarding school in Kendal, he moved on to a teaching position in the burgeoning city of Manchester. There he joined the manchester literary and philosophical society, which provided him with a stimulating intellectual environment and laboratory facilities. The first paper he delivered before the society was on color blindness, which afflicted him and is sometimes still called daltonism.
Ernest. Rutherford • At Manchester, Rutherford continued his research on the properties of the radium emanation and of the alpha rays and, in conjunction with H. Geiger, a method of detecting a single alpha particle and counting the number emitted from radium was devised. In 1910, his investigations into the scattering of alpha rays and the nature of the inner structure of the atom which caused such scattering led to the postulation of his concept of the "nucleus", his greatest contribution to physics. According to him practically the whole mass of the atom and at the same time all positive charge of the atom is concentrated in a minute space at the centre. In 1912 Niels Bohr joined him at Manchester and he adapted Rutherford's nuclear structure to Max Planck's quantum theory and so obtained a theory of atomic structure which, with later improvements, mainly as a result of Heisenberg's concepts, remains valid to this day.
Ernest. Rutherford • In 1913, together with H. G. Moseley, he used cathode rays to bombard atoms of various elements and showed that the inner structures correspond with a group of lines which characterize the elements. Each element could then be assigned an atomic number and, more important, the properties of each element could be defined by this number. In 1919, during his last year at Manchester, he discovered that the nuclei of certain light elements, such as nitrogen, could be "disintegrated" by the impact of energetic alpha particles coming from some radioactive source, and that during this process fast protons were emitted. Blackett later proved, with the cloud chamber, that the nitrogen in this process was actually transformed into an oxygen isotope, so that Rutherford was the first to deliberately transmute one element into another. G. de Hevesy was also one of Rutherford's collaborators at Manchester
Niels Bohr • After matriculation at the Gammelholm Grammar School in 1903, he entered Copenhagen University where he came under the guidance of Professor C. Christiansen, a profoundly original and highly endowed physicist, and took his Master's degree in Physics in 1909 and his Doctor's degree in 1911.
Niels Bohr • While still a student, the announcement by the Academy of Sciences in Copenhagen of a prize to be awarded for the solution of a certain scientific problem, caused him to take up an experimental and theoretical investigation of the surface tension by means of oscillating fluid jets. This work, which he carried out in his father's laboratory and for which he received the prize offered (a gold medal), was published in the Transactions of the Royal Society, 1908.
John. Thomson • In 1896, Thomson visited America to give a course of four lectures, which summarised his current researches, at Princeton. These lectures were subsequently published as Discharge of Electricity through Gases (1897). On his return from America, he achieved the most brilliant work of his life - an original study of cathode rays culminating in the discovery of the electron, which was announced during the course of his evening lecture to the Royal Institution on Friday, April 30, 1897. His book, Conduction of Electricity through Gases, published in 1903 was described by Lord Rayleigh as a review of "Thomson's great days at the Cavendish Laboratory". A later edition, written in collaboration with his son, George, appeared in two volumes (1928 and 1933).
John. Thomson • His book, Conduction of Electricity through Gases, published in 1903 was described by Lord Rayleigh as a review of "Thomson's great days at the Cavendish Laboratory". A later edition, written in collaboration with his son, George, appeared in two volumes (1928 and 1933).
Nucleus • The nuclear dimension is in the range of 10-13 10-12 cm, while the atomic dimension is about 10-8 cm. That is, the diameter of an atom is over 10,000 times the diameter of its nucleus • This nuclear range is called a short-range
Molecules • Molecules are small particles that make up all living and non-living things. They are made up of even tinier particlescalled atoms. Molecules in living thingsare made from only about 25 of more than 100 known atoms in the universe. Moleculesare made from as few as two atoms to hundredsof millions of atoms.
Mass number <3 • The mass number (A), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus
Isotope • Atoms of the same element can have different numbers of neutrons; the different possible versions of each element are called isotopes. For example, the most common isotope of hydrogen has no neutrons at all; there's also a hydrogen isotope called deuterium, with one neutron, and another, tritium, with two neutrons.
Subatomic particles • It is a spin-half lepton that participates in electromagnetic interactions, and its mass is less than one thousandth of that of the smallest atom. • Its electric charge is defined by convention to be negative, with a value of -1 in atomic units. • Together with atomic nuclei, electrons make up atoms; their interaction with adjacent nuclei is the main cause of chemical bonding.
Subatomic Particles • Its electric charge is defined by convention to be negative, with a value of -1 in atomic units. • Together with atomic nuclei, electrons make up atoms; their interaction with adjacent nuclei is the main cause of chemical bonding.
Subatomic Particles • The proton is a subatomic particle with an electric charge of one positive fundamental unit and a mass of 938.3 MeV/c2, or about 1836 times the mass of an electron.
Subatomic Particles • The proton is observed to be stable, with a lower limit on its half-life of about 1035 years, although some theories predict that the proton may decay.
Subatomic Particles • Neutrons have no electrical charge and are said to help hold the protons together (protons are positively charged particles and should repel each other). If all protons are identical and all neutrons are identical, then what makes the atoms of two different elements different from each other? For example, what makes a hydrogen atom different from a helium atom? The number of protons and neutrons in the nucleus give the atoms their specific characteristics. In the graphic below you will notice that each of the three elements have different numbers of protons and neutrons. They would also like to have the same number of electrons as they have protons in order to stay electrically balanced.
Spectrograph Monochromator and spectrograph systems form an image of the entrance slit in the exit plane at the wavelengths present in the light source. There are numerous configurations by which this may be achieved - only the most common are discussed in this document and includes Plane Grating Systems (PGS) and Aberration Corrected Holographic Grating (ACHG) systems. Definitions LA - entrance arm length LB - exit arm length h - height of entrance slit h' - height of image of the entrance slit α - angle of incidence β - angle of diffraction w - width of entrance slit w' - width of entrance slit image Dg - diameter of a circular grating Wg - width of a rectangular grating Hg - height of a rectangular grating
Periodic Tables <3 • Periods • Rows of elements are called periods. The period number of an element signifies the highest unexcited energy level for an electron in that element. The number of elements in a period increases as you move down the periodic table because there are more sublevels per level as the energy level of the atom increases
Periodic Table…Chemical Symbol • Ac Actinium 89 • - Atomic number Ag Silver 47 • - Symbol Al Aluminum 13 • - Atomic Mass Am Americium 95 • - Electronegativity Ar Argon 18 • - Density As Arsenic 33 • - Melting point At Astatine 85 • - Boiling point Au Gold 79 • - Vanderwaals radius B Boron 5 • - Year of discovery Ba Barium 56 • - Inventor surname Be Beryllium 4 • - Elements in earthcrustBhBohrium 107 • - Elements in human body Bi Bismuth 83 • - Covalenz radius Bk Berkelium 97 • - Ionization energy Br Bromine 35 • For chemistry students and teachers: The tabular chart on the right is arranged by the symbol. • The first chemical element is Actinium and the last is Zirconium. • Please note that the elements do not show their natural relation towards each other as in the Periodic system. There you can find the metals, semi-conductor(s), non-metal(s), inert noble gas(ses), Halogens, Lanthanoides, Actinoids (rare earth elements) and transition metals. • C Carbon 6 • Ca Calcium 20 • Cd Cadmium 48 • Ce Cerium 58 • Cf Californium 98 • Cl Chlorine 17 • Cm Curium 96 • Co Cobalt 27
Periodic Table…Chemical Symbol • Cr Chromium 24 • Cs Cesium 55 • Cu Copper 29 • DbDubnium 105 • Ds Darmstadtium 110 • Dy Dysprosium 66 • Er Erbium 68 • EsEinsteinium 99 • Eu Europium 63 • F Fluorine 9 • Fe Iron 26 • Fm Fermium 100 • Fr Francium 87 • Ga Gallium 31 • Gd Gadolinium 64 • Ge Germanium 32 • H Hydrogen 1 • He Helium 2 • Hf Hafnium 72 • Hg Mercury 80 • Ho Holmium 67 • HsHassium 108 • I Iodine 53 • In Indium 49 • Ir Iridium 77 • K Potassium 19 • Kr Krypton 36 • La Lanthanum 57 • Li Lithium 3 • Lr Lawrencium 103 • Lu Lutetium 71 • Md Mendelevium 101 • Mg Magnesium 12 • Mn Manganese 25 • Mo Molybdenum 42 • Mt Meitnerium 109 • N Nitrogen 7 • Na Sodium 11 • Nb Niobium 41 • Nd Neodymium 60 • Ne Neon 10 • Ni Nickel 28 • No Nobelium 102
Periodic Table…Chemical Symbol • Np Neptunium 93 • O Oxygen 8 • Os Osmium 76 • P Phosphorus 15 • Pa Protactinium 91 • Pb Lead 82 • Pd Palladium 46 • Pm Promethium 61 • Po Polonium 84 • Pr Praseodymium 59 • Pt Platinum 78 • Pu Plutonium 94 • Ra Radium 88 • Rb Rubidium 37 • Re Rhenium 75 • RfRutherfordium 104 • RgRoentgenium 111 • Rh Rhodium 45 • Rn Radon 86 • Ru Ruthenium 44 • S Sulfur 16 • Sb Antimony 51 • Sc Scandium 21 • Se Selenium 34 • SgSeaborgium 106 • Si Silicon 14 • Sm Samarium 62 • Sn Tin 50 • Sr Strontium 38 • Ta Tantalum 73 • Tb Terbium 65 • Tc Technetium 43 • Te Tellurium 52 • Th Thorium 90 • Ti Titanium 22 • Tl Thallium 81 • Tm Thulium 69 • U Uranium 92 • UubUnunbium 112 • UuhUnunhexium 116 • UuoUnunoctium 118 • UupUnunpentium 115 • UuqUnunquadium 114
Periodic Table…Chemical Symbol • UusUnunseptium 117 • UutUnuntrium 113 • V Vanadium 23 • W Tungsten 74 • Xe Xenon 54 • Y Yttrium 39 • Yb Ytterbium 70 • Zn Zinc 30 • Zr Zirconium 40
Electron shells • In atomic physics and quantum chemistry, electron configurationis the arrangement of electrons of an atom, a molecule, or other physical structure.[1] It concerns the way electrons can be distributed in the orbitals of the given system (atomic or molecular for instance).
Neutral atom • The interaction between charged and neutral particles is a common phenomenon in space plasmas. Whenever an energetic ion undergoes a charge exchange process in a collision with a neutral background atom, an energetic neutral atom – ENA – is born. Ion-electron recombination and neutral atom acceleration by the solar gravitation may also contribute to an ENA population under certain conditions. ENAs are ubiquitous in space environment and their study opens a new window on various phenomena in space plasmas with a promise (already partially realized) to qualitatively improve our understanding of global magnetospheric and heliospheric processes
Reactivity • It is useful to begin a discussion of organic chemical reactions with a review of acid-base chemistry and terminology for several reasons. First, acid-base reactions are among the simplest to recognize and understand. Second, some classes of organic compounds have distinctly acidic properties, and some other classes behave as bases, so we need to identify these aspects of their chemistry. Finally, many organic reactions are catalyzed by acids and/or bases, and although such transformations may seem complex, our understanding of how they occur often begins with the functioning of the catalyst.
Reactivity • Organic chemists use two acid-base theories for interpreting and planning their work.
:D All Done!!! <3 • By Emily <3