Molar Conversions
Unit 6 - The Mole. Molar Conversions. VERY. A large amount!!!!. A. What is the Mole?. A counting number (like a dozen) Avogadro’s number (N A ) 1 mol = 6.02 10 23 items. A. What is the Mole?. HOW LARGE IS IT???.

Molar Conversions
E N D
Presentation Transcript
Unit 6 - The Mole Molar Conversions
VERY A large amount!!!! A. What is the Mole? • A counting number (like a dozen) • Avogadro’s number (NA) • 1 mol = 6.02 1023 items
A. What is the Mole? HOW LARGE IS IT??? • 1 mole of pennies would cover the Earth 1/4 mile deep! E.C. ($) • 1 mole of hockey pucks would equal the mass of the moon! • 1 mole of basketballs would fill a bag the size of the earth!
EC Hints • 5.1×108 km2 = surface area of Earth • Find diameter and width of penny in mm • 0.25 miles = __?__ mm • How much $$$ do we have here???
Mole Humor • Q: What is a mole's favorite movie? The Green Mole • Q: What did Avogadro teach his students in math class? Moleteplication • Why was there only one Avogadro? When they made him, they broke the Moled
B. Molar Mass • Mass of 1 mole of an element or compound. • Atomic mass tells the... • atomic mass units per atom (amu) • grams per mole (g/mol) • Round to 2 decimal places (hundredths; ie: 16.01 )
B. Molar Mass Examples 12.01 g/mol 26.98 g/mol 65.39 g/mol • carbon • aluminum • zinc
B. Molar Mass Examples • water • sodium chloride • H2O • 2(1.01) + 16.00 = 18.02 g/mol • NaCl • 22.99 + 35.45 = 58.44 g/mol
B. Molar Mass Examples • sodium bicarbonate • sucrose • NaHCO3 • 22.99 + 1.01 + 12.01 + 3(16.00) = 84.01 g/mol • C12H22O11 • 12(12.01) + 22(1.01) + 11(16.00) = 342.34 g/mol
Mole Humor • What did Avogadro invent for his wife to use as a night cream? Oil of Molay • How much does Avogadro exaggerate? He makes mountains out of mole hills • What is Avogadro's favorite kind of music? Rock 'N' Mole
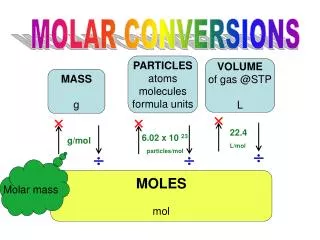
MASS IN GRAMS MOLES NUMBER OF PARTICLES C. Molar Conversions molar mass 6.02 1023 (g/mol) (particles/mol)
C. Molar Conversion ExamplesGramsMoles • How many moles of carbon are in 26 g of carbon? 26 g C 1 mol C 12.01 g C = 2.2 mol C
C. Molar Conversion ExamplesMolesGrams • How many grams are in 21.3 moles of CO2 21.3 moles CO2 44.01 g 1mol CO2 = 937 g CO2
C. Molar Conversion ExamplesMolesMolecules • How many molecules are in 2.50 moles of C12H22O11? 6.02 1023 molecules 1 mol 2.50 mol = 1.51 1024 molecules C12H22O11
C. Molar Conversion ExamplesMoleculesMoles • Convert 1.75 x 1026 atoms of K to moles of K. 1 mol 6.02 1023 molecules K 1.75 x 1026 atoms K = 291 mol K
C. Molar Conversion ExamplesMoleculesGrams • Find the mass of 2.1 1024 molecules of NaHCO3. 2.1 1024 molecules 1 mol 6.02 1023 molecules 84.01 g 1 mol = 290 g NaHCO3
Unit 6 – The Mole III. Formula Calculations
A. Percentage Composition • the percentage by mass of each element in a compound
127.10 g Cu 159.17 g Cu2S 32.07 g S 159.17 g Cu2S A. Percentage Composition • Find the % composition of Cu2S. 100 = %Cu = 79.852% Cu %S = 100 = 20.15% S
CH3 B. Empirical Formula • Smallest whole number ratio of atoms in a compound C2H6 reduce subscripts C. Johannesson
C2H6 C. Molecular Formula • “True Formula” - the actual number of atoms in a compound CH3 empirical formula ? molecular formula