Chapter 19 Oxidative Phosphorylation
641 likes | 1.22k Views
Chapter 19 Oxidative Phosphorylation. Chapter 14. Chapter 16. Chapter 19. Electron transport and oxidative phosphorylation are membrane-associated processes. Bacteria---- electron transport and oxidative phosphorylation are carried out at and across plasma membrane

Chapter 19 Oxidative Phosphorylation
E N D
Presentation Transcript
Chapter 19 Oxidative Phosphorylation
Chapter 14 Chapter 16 Chapter 19
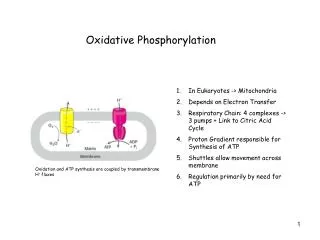
Electron transport and oxidative phosphorylation are membrane-associated processes • Bacteria---- electron transport and oxidative phosphorylation are carried out at and across plasma membrane • Eukaryotic cells---mitochondria • (Plants---chloroplasts)
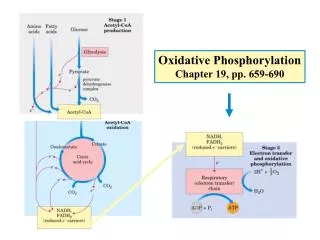
Mitochondria 1. TCA, fatty acid oxidation, electron transport and oxidative phosphorylation occur in mitochondria 2. Mammalian contains 800 to 2500 mitochondria, some types of cells have one or two, and some have as many as half million, human erythrocytes have none. 3. Outer membrane, intermembrane space, inner membrane, cristae, matrix 4. Genetic materials
Permeable to Mr. 5,000 Porin 37 genes, 13 encoding ETC proteins (most mitochondrial proteins are encoded by nucleus and then transported into mitochondria)
Electrons are funneled to universal electron acceptors • Flavoproteins---FAD or FMN containing proteins (see below) • NAD-linked dehydrogenase and NADP-linked dehydrogenase Reduced substrate+NAD+→Oxidized substrate+NADH + H+ Reduced substrate+NADP+→Oxidized substrate+NADPH + H+
NADH & NADPH • Not metabolically interchangeable • NADH : use the free energy of metabolite oxidation to synthesize ATP • NADPH : use the free energy of metabolite oxidation for otherwise energy endergonic reductive biosynthesis
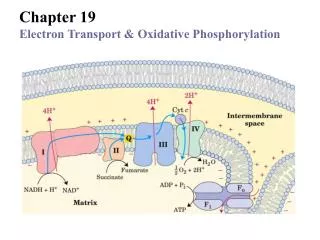
The Electron Transport Chain—An Overview The electron transport chain can be isolated in four complexes---- Complex I: NADH::Ubiquinone oxidoreductase, Complex II:Succinate dehydrogenase, Complex III:ubiquinone-cytochrome c oxidoreductase Complex IV:cytochrome c oxidase
Electron Carriers • Flavoprotein--Tightly bound FMN or FAD, one or two e- carrier • Coenzyme Q, also called ubiquinone (abbr. CoQ or Q)--one or two e- carrier, mobile within membrane • cytochromes--- Fe2+/Fe3+, one e- carrier • cytochrome a’s: Isoprenoid (15-C) on modified vinyl and formyl in place of methyl • cytochrome b’s: Iron-protoporphyrin IX • cytochrome c’s: Iron-protoporphyrin IX linked to cysteine • iron-sulfur proteins--- one e- carrier, several types • coppers--- one e- transfer, Cu+/Cu++
Prosthetic groups of cytochromes Fig. 19-3
Iron-sulfur centers Fe 2Fe-2S 4Fe-4S
Determining the sequence of electron flow • Standard reduction potential (table 19-2) • Reducing the entire chain of carrier experimentally by providing an electron source but no electron acceptor (no O2). • Inhibitors of electron transport chains. Page 1 of 3
Reduction potentials • Redox couples that have large positive reduction potential have a strong tendency to accept electrons, and the oxidized form of such a couple is a strong oxidizing agent. • Redox couples with large negative reduction potentials have a strong tendency to undergo oxidation, and the reduced form of such a couple is a strong reducing agent.
Determining the sequence of electron flow • Standard reduction potential (table 19-2) • Reducing the entire chain of carrier experimentally by providing an electron source but no electron acceptor (no O2). • Inhibitors of electron transport chains. Page 2 of 3
Reducing the entire chain of carrier experimentally by providing an electron source but no electron acceptor (no O2). When O2 is suddenly introduced into the system, the rate at which each electron carriers oxidized (measured spectroscopically) shows the order in which the carriers function. 2. The carrier nearest O2 (at the end of the chain) gives up its electrons first, the second carrier from the end is oxidized next, and so on.
Determining the sequence of electron flow • Standard reduction potential (table 19-2) • Reducing the entire chain of carrier experimentally by providing an electron source but no electron acceptor (no O2). • Inhibitors of electron transport chains. Page 3 of 3
FIGURE 19–6 Method for determining the sequence of electron carriers. This method measures the effects of inhibitors of electron transfer on the oxidation state of each carrier. In the presence of an electron donor and O2, each inhibitor causes a characteristic pattern of oxidized/reduced carriers: those before the block become reduced (blue), and those after the block become oxidized (pink).
Electron carriers function in multienzyme complexes The electron transport chain can be isolated in four complexes---- Complex I: NADH::Ubiquinone oxidoreductase, Complex II:Succinate dehydrogenase, Complex III:ubiquinone-cytochrome c oxidoreductase Complex IV:cytochrome c oxidase
Complex I: NADH::Ubiquinone oxidoreductase, (NADH dehydrogenase) • Transfer one pair of e- from NADH to coenzyme Q • Reaction steps: (1) transfer 2e- from NADH to [FMN] (2) transfer e- from [FMNH2] to Fe-S cluster, (3) transfer e- from Fe-S cluster to coenzyme Q. • Four H+ transported per 2 e- passes from NADH to UQ. • Coenzyme Q is a mobile electron carrier which shuttles e- from Complexes I and II to III. • P (for positive, intermembrane space) face and N (for negative, matrix) face • Protons pumped from matrix to cytosol
Fig. 19-9 Inhibitors: Amytal, rotenone and pieridin A
Complex II: succinate-coenzyme Q reductase (succinate dehydrogenase) • The only TCA cycle enzyme that is an integral membrane protein, also called flavoprotein (FP2). • Succinate →fumarate + 2H+ + 2e- UQ + 2H + + 2e- → UQH2 • Components: FAD and Fe-S centers • No proton pumped • Similar complexes • Glycerolphosphate dehydrogenase: Reduces CoQ: No protons pumped • Fatty acyl-CoA dehydrogenase:Reduces CoQ: No protons pumped
Complex III: Coenzyme Q-cytochrome c reductase • CoQ passes electrons to cyt c (and pumps H+) in a unique redox cycle known as the Q cycle • The principal transmembrane protein in complex III is the b cytochrome - with hemes bL and bH • Cytochromes, like Fe in Fe-S clusters, are one- electron transfer agents • Study Figure 19.11 - the Q cycle • UQH2 is a lipid-soluble electron carrier • cyt c is a water-soluble electron carrier
Q cycle • First half reaction: A molecule UQH2 diffuses to Qp site on Complex III, an e- from UQH2 is transferred to the Rieske protein and then to cytochrome c1. This releases two H+ to the cytosol and leaves UQ.-. The second e- is then transferred to the bL heme, converting UQ.- to UQ: • Second half reaction: oxidation of UQH2 at QN site, one e- being passed to cytochrome c1 and the other transferred to heme bL and then to bH.
The Q cycle Fig. 19-12
Complex IV: cytochrome c oxidase • Electrons from cytc are used in a four-electron reduction of O2 to produce 2H2O • Oxygen is thus the terminal acceptor of electrons in the electron transport pathway - the end! • Cytochrome c oxidase utilizes 2 hemes (a and a3) and 2 copper sites • Structure is now known - mostly! • Complex IV also transports H+
Path of e- through complex IV Fig. 19-14
Summary of the flow of electrons and protons through the four complexes of the respiratory chain Fig. 19-15
Coupling e- Transport and Oxidative Phosphorylation (ATP synthesis) • This coupling was a mystery for many years • Many biochemists squandered careers searching for the elusive "high energy intermediate" • Peter Mitchell proposed a novel idea - a proton gradient across the inner membrane could be used to drive ATP synthesis • Mitchell was ridiculed, but the chemiosmotic hypothesis eventually won him a Nobel prize • Be able to calculate the G for a proton gradient (Figure 19-15)
Chemiosmotic model Fig. 19-17
Mitchell’s chemiosmotic hypothesis • Proton gradient used to drive ATP synthesis • Protons per electron pair • From succinate 6 • From NADH 10 • Four protons per ATP • ADP uptake: 1 proton • ATP synthesis: 3 protons • One oxygen atom consumed per electron pair • P/O: the number of ATP produced per atom of oxygen consumed. Used as an index of oxidative phosphorylation. • From NADH: 2.5 • From succinate: 1.5
P/O ratio • ADP + Pi ATP • P/O: the number of ATP produced per atom of oxygen consumed. Used as an index of oxidative phosphorylation. • From NADH: 2.5 ATP • From succinate: 1.5 ATP
ATP synthase Proton diffusion through the protein drives ATP synthesis! • Two parts: F1 and F0 (latter was originally "F-o" for its inhibition by oligomycin) ---F1: ATP synthesis, a3b3gde ---Fo: Proton channel in inner membrane, ab2c10-12 • Binding change mechanism: the three catalytic b subunits are intrinsically identical but are not functionally equivalently at any particular moment where they cycle through three conformational states. • 18O exchange experiment: the role of proton gradient is not to form ATP but to release it from the enzyme
Mitochondrial ATP synthase complex Fig. 19-23
Mitochondrial ATP synthase complex Fig. 19-23
Catalytic mechanism of F1:18O exchange experiment Figure 19-21
Binding change model for ATP synthesis Fig. 19-24
Adenine nucleotide and phosphate translocase • ATP must be transported out of the mitochondria • Adenine nucleotide translocase • ATP-ADP antiporter located in inner mitochondrial membrane • But ATP4- out and ADP3- in is net movement of a negative charge out - equivalent to a H+ going in • The proton-motive force drives ATP-ADP exchange • Phosphate translocase • Symport of H2PO4- and H+ located in inner mitochondrial membrane • Consume some of the energy of e- transport (H+ moved from P to the N side of the inner membrane)
There is no net flow of charge during symport of H2PO4 and H+ Fig. 19-26
Shuttle Systems for e - • Most NADH used in electron transport is cytosolic and NADH doesn't cross the inner mitochondrial membrane • What to do?? • "Shuttle systems" effect electron movement without actually carrying NADH • Glycerol 3-phosphate shuttle stores electrons in glycerol-3-P, which transfers electrons to FAD • Malate-aspartate shuttle uses malate to carry electrons across the membrane
Malate aspartate shuttle Liver, kidney and heart Fig. 19-27