Elements,Compounds and Mixtures
Elements,Compounds and Mixtures. Mixtures. Consist of a physical blend of two or more substances. They contain a variable composition Most materials found in nature are mixtures. EX. Air, stew, blood. Mixtures.

Elements,Compounds and Mixtures
E N D
Presentation Transcript
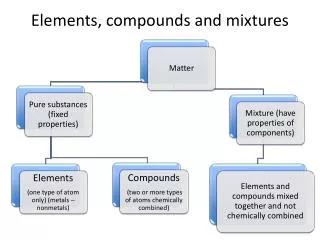
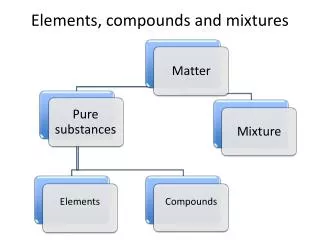
Mixtures • Consist of a physical blend of two or more substances. • They contain a variable composition • Most materials found in nature are mixtures. • EX. Air, stew, blood
Mixtures • The creation of a mixture results from a physical change where no new substances are created. Some mixtures can be separated by physical methods. Example: Sulfur and Iron fillings with a magnet. • Mixtures can be homogeneous or heterogeneous.
Mixtures • Heterogeneous mixtures are not uniform in composition.Ex. soil • Homogeneous mixtures are uniform in composition. • Ex. Tooth paste
Elements,Compounds,Mixtures • Every substance is either an element or a compound. • Elements are the simplest form of matter. • Elements are the building blocks for all other substances.
ELEMENTS • Simple pure substances • all atoms the same • can’t be divided by physical or chemical means • most solids at room temperature. • some in free state but most combined with other elements
3 classes of elements • Metals • solids, malleable, ductile, good conductors, mercury the only liquid at RT. • Nonmetals • solids and gases, brittle, poor conductors • bromine the only liquid at RT. • Metalloids • share properties of both • Ex.Si, Ge, As, Sb, Te
Symbols of Elements • One kind of atom • represented by 1, 2 or 3 letters • carbon = C (1st letter) • calcium = Ca (1st 2 letters) • magnesium = Mg ( 1st and 3rd letter) • iron = Fe (from Latin name) • tungsten = W (from German name wolfram)
substances Pure substances mixtures hetero homo elements compounds ionic molecular alloys solutions metal nonmetal
Law of conservation of energy • Energy can be converted from one form to another but can’t be created or destroyed • Potential energy • stored, energy of position, bond energy • Kinetic energy • energy in motion
Law of conservation of mass • There is never a loss of mass in a chemical reaction • 30g of reactants will produce 30g of products if combined in the correct ratio • ppt = precipitate • a solid that forms when 2 solutions are mixed
Iron and Sulfur • The reaction of sulphur
Iron and Sulfur • ...with iron is often used to show the differences between elements, compounds and mixtures and between chemical and physical changes.
Iron and Sulfur • This picture shows the mixture of iron and sulfur.
Iron and Sulfur • In this picture a chemical change is taking place. When the elements iron and sulfur are being heated, they combine to form the compound iron(II) sulfide. This reaction is typical of the way in which sulfur reacts with many metals. The equation for the reaction is: • Fe + S -> FeS
Iron and Sulfur • This is the resulting compound of iron sulfide. The heating has produced a chemical change.