ELEMENTS, COMPOUNDS AND MIXTURES
150 likes | 346 Views
ELEMENTS, COMPOUNDS AND MIXTURES. Food Chemistry. OBJECTIVES. Distinguish between pure substances and mixtures. Compare homogeneous and heterogeneous mixtures. Define solutions. Distinguish between a solute and solvent. Recognize water as a universal solvent.

ELEMENTS, COMPOUNDS AND MIXTURES
E N D
Presentation Transcript
ELEMENTS, COMPOUNDS AND MIXTURES Food Chemistry
OBJECTIVES • Distinguish between pure substances and mixtures. • Compare homogeneous and heterogeneous mixtures. • Define solutions. • Distinguish between a solute and solvent. • Recognize water as a universal solvent. • Understand the processes of solubility and saturation.
Overview Matter • Anything that has mass and takes up space. Physical Properties • A characteristic that can be observed or measured without changing the substance • Solid • Liquid • Gas Chemical Properties • The ability of a substance to react with other substances
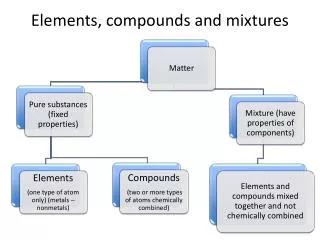
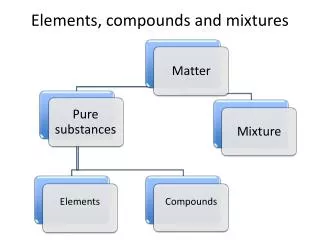
Classifications of matter www3.delta.edu
Pure substances • Made of only one kind of material and has definite properties Elements • Sodium, Carbon, Iron Compounds • Table salt, water, sugar
Mixtures • A combination of two or more substances in which each substance keeps at least some of its original properties. • Physically blended, not chemically. Homogeneous Mixtures Heterogeneous Mixtures Solutions Colloids
Homogeneous mixtures • The same in every part of a given sample. Examples: salt water, milk, air, cola Another name for a homogeneous mixture: Solution – one substance is dissolved in another Solution Solute Solvent
Solute vs. solvent • The substance that is dissolved • Sugar • Salt • The substance that dissolves another substance • Water Water is recognized as a universal solvent. This can be a problem with vitamins and minerals dissolving in cooking water. SOLUTE SOLVENT
Heterogeneous Mixtures • Individual substances are dissimilar and can be recognized by sight. This is also known as “Immiscible” Examples: salad, potato soup, Italian dressing
Saturated vs. Unsaturated solutions Unsaturated Solution: • A solution that contains less solute than can be dissolved in it at a given temperature. Saturated Solution: • A solution that contains all the solute that can be dissolved at a given temperature.
solubility • The maximum amount of solute that can dissolve in a given quantity of solvent at a specific temperature. • The solubility of most solids dissolved in a liquid increases as the temperature increases. Example: Sugar dissolves faster in boiling water. • The solubility of gas decreases as the temperature increases. Example: A warm can of soda will go flat faster than a cold can of soda.
Solute & phase changes • The amount of solute in a solution affects the temperature at which the solution boils or freezes. • The more solute present in a solution, the higher the boiling point and the lower the freezing point. Example: Ice cream is still soft in the freezer because of the sugar dissolved in the water.
Identify the mixtures Your table now has 4 glasses. Add 4 different solutions to the glasses and identify the type of mixture as homogeneous or heterogeneous. Glass # 1 – Place 1 drop of food coloring in your glass of water and stir. Glass # 2 – Add ¼ cup oil to the water. Glass # 3 – Add ¼ cup vinegar the water. Glass # 4 – Add ¼ cup vinegar to the oil.
Solubility Lab • Make Iced Tea using both cold and hot water. • Compare the flavors and answer the questions to review the scientific theme of the week.