Heat Transfer
Heat Transfer . Schindewolf 8 th grade Science. Objectives…. What do you think heat is? How does heat transfer from one thing to another? How come some things get hotter than others? These are the three things we are going to learn today!. Objective #1: “What IS heat?”.

Heat Transfer
E N D
Presentation Transcript
Heat Transfer Schindewolf 8th grade Science
Objectives… • What do you think heat is? • How does heat transfer from one thing to another? • How come some things get hotter than others? • These are the three things we are going to learn today!
Objective #1: “What IS heat?” • There are 2 things we need to know BEFORE we can define heat! • “Temperature” and “Thermal Energy” • Let’s start with temperature: What do you think “temperature” is?
Temperature • Remember: EVERYTHING is made up of tiny particles called “atoms” in constant motion! • The they move the more “kinetic energy” they have. (Remember Kinetic energy?- The energy of motion) • Temperature is “A measure of the average Kinetic Energy of INDIVIDUAL particles!” faster
Temperature Scales Hot Cool • There are three main scales to measure temperature. • Used mainly in the US. Freezing is 32 degrees Boiling is 212 degrees Farenheit F
Temperature Scales Hot Cool • The “metric” temperature scale. • Used everywhere else in the world. Freezing is 0 degrees Boiling is 100 degrees Celsius C
Temperature Scales Hot Cool • Used mainly by Physicists. • Kelvin is Celsius + 273 degrees. Freezing is 273 degrees Boiling is 373 degrees “That seems silly! WHY do we need this?” The lowest possible temperature EVER recorded was -273 degrees Cesius! Scientists therefore created a temperature scale where 0 would reflect the coldest possible temperature. Thus Kelivn was born with it’s “absolute zero” Kelvin BRRR K
Thermal Energy • What was temperature? “ A measure of the average Kinetic Energy of INDIVIDUAL particles” • Thermal Energy is the “TOTAL energy of ALL the particles in a substance.” KE of particle 1 + KE of particle 2 + KE of particle 3 + KE of particle 4…….. (You get the idea)
Thermal Energy AVERAGE energy of particles is = for both cups! TOTAL energy of ALL particles is greater for enormo-cup because there is a greater number of particles! Consider a small cup of hot chocolate and a big cup of hot chocolate. The BIG cup has the same “Temperature” (average Kinetic Energy of particles. However it has a lot greater “Thermal Energy” (energy of ALL the particles combined!)
So what does this have to do with heat? • Now that we have defined Temperature and Thermal Energy we are ready to define HEAT! • Heat is “The transfer of thermal energy from an object at one temperature to an object at another temperature.” Objective #1 “What is Heat?”
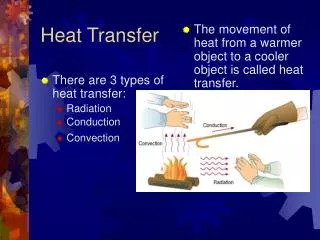
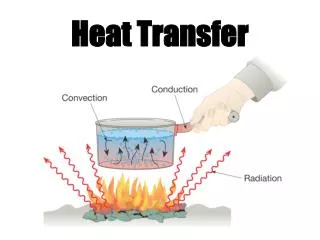
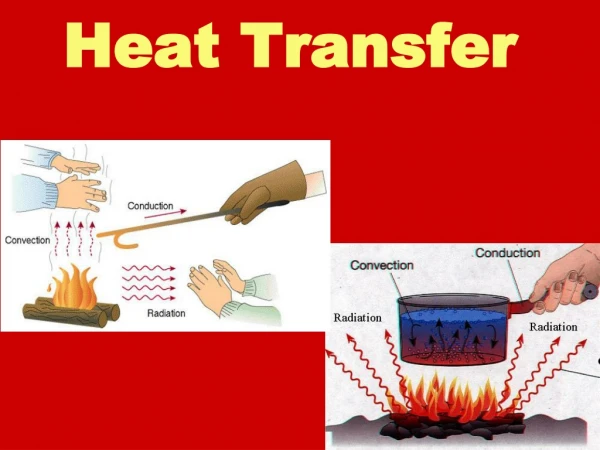
Objective #2: “How does heat transfer from one object to another?” • How does heat get from the sun to us? Or from your stove top to your pan? • It’s time to meet the three “heat transfer” words: Radiation Convection Conduction
First: Let’s learn about Conduction • Conduction is the easy one to understand! • Conduction happens when one object touches another object and transfers heat. hot
Think “CONTACT” Conduction • Let’s talk about examples of conduction “transfer by direct contact” • The bottom of a metal pot on a stovetop! A metal spoon heats up when held inside the flame of a bunsen burner and a candle melts when touched to a hot spoon “Remember that?” Contact
Next: Let’s learn about Convection • Convection’s key word is “currents” in liquids or gasses! • Convection happens when movement of hot/cold particles in a solid or a gas creates a current.. • Consider a pot of boiling water… • Remember hot water is less dense than cold water? Hot water rises as cold water sinks. When the cold water that has “sunk” to the bottom is heated, it rises back to the top. The result is a constant current where all the particles are heated. Hot Water Cold Water Hot Water Cold Water Is heated
Think “Current” Convection • Let’s talk about examples of convection “heat transfer by “current” • The air flow in a room… • Water boiling in a pot. Currents
Last type of heat transfer: Radiation • Radiation’s key word is • Radiation is the transfer of energy by electromagnetic waves! • Radiation is unique because it is the ONLY type of heat transfer that does not require matter. (Like the Sun’s rays, it can travel even through space!” waves
Radiation Think WAVES • Let’s talk about examples of Radiation. • A toaster! • A Microwave! • The Sun! • A Heat Lamp (like the one we used to heat sand and water!) WAVES Objective#2 “How does heat transfer?”
Objective #3 “How come some things get hotter than others” • Have you ever been to the beach and run across burning hot sand? • Was the water equally as hot? Or did it feel cool and refreshing? • Both objects were exposed to the same amount of “Radiation” from the Sun. • So why is one so much Ouch! hotter?
The answer is…… • Specific Heat • The Specific Heat of a substance tells how much energy is needed to raise the temperature of 1 KG of a substance 1 degree Kelvin. Raise 1KG 1 Degree K
Specific Heat • Specific Heat is kind of like a price. • Sand has a specific heat of 670. This means it “charges” 670 Joules to raise 1Kg of Sand 1 degree K! • Water has a specific heat of 4, 180. This means it “charges” 4, 180 Joules to raise 1Kg of Water 1 degree K.
Specific Heat • Sand = 670J Water = 4,180J • Sand therefore heats up much much faster than water does given the same amount of energy! • The the specific heat, the it heats up! faster Objective #3 “Why do some objects heat up faster than others?
PRACTICE! • Try this game and see how you do!