ENZYMES
PTT 202: ORGANIC CHEMISTRY FOR BIOTECHNOLOGY. ENZYMES. PREPARED BY: NOR HELYA IMAN KAMALUDIN helya@unimap.edu.my. Introduction of enzymes. Enzyme in biological reactions.

ENZYMES
E N D
Presentation Transcript
PTT 202: ORGANIC CHEMISTRY FOR BIOTECHNOLOGY ENZYMES PREPARED BY: NOR HELYA IMAN KAMALUDIN helya@unimap.edu.my
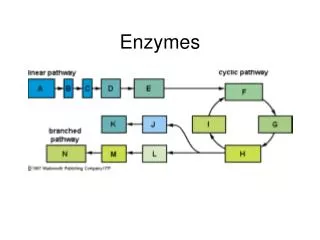
Enzyme in biological reactions Enzymes are "biological catalysts." "Biological" means the substance that is produced or is derived from some living organism. "Catalyst" denotes a substance that has the ability to increase the rate of a chemical reaction, and is not changed or destroyed by the chemical reaction that it accelerates. Catalysts are specific in nature as to the type of reaction they can catalyze. Enzymes, as a subclass of catalysts, are very specific in nature. Each enzyme can act to catalyze only very select chemical reactions and only with very select substances. Many chemical reactions do proceed but at such a slow rate that their progress would seem to be imperceptible at normally encountered environmental temperature Enzymes often increase the rate of a chemical reaction between 10 and 20 million times what the speed of reaction would be when left uncatalyzed (at a given temperature).
Catalytic antibodies Catalytic antibodies is the ability of the highly evolved machinery of immune system to produce structurally and functionally complex molecules like antibodies The idea of using antibodies to catalyze chemical reactions can be traced back to the catalytic concept of Linus Pauling. According to Pauling, if the structure of the antigen binding site of antibodies were to be produced in a random manner, the antigen binding site of some of the antibodies may resemble the active site of enzymes and such molecules could have the ability to transform substrates to products.
Coupled assay method Example of coupled assay reactions Indicator enzyme Auxiliary enzyme
Measurement of enzyme activity In order to follow the progress of an enzyme-catalysed reaction, it is necessary to measure either the depletion of the substrate or the accumulation of the product. The rate at which the substrate is converted to the product by the action of an enzyme is dependent upon the concentrations of both the enzyme and the substrate. Figure 1: The initial rate of such a reaction is maximal and it is the initial rate of reaction that reflects to the enzyme activity.
Measurement of enzyme activity • The rate of reaction declines from a maximal initial velocity, V0, which can be represented by the tangent to the curve at zero time. The rate falls least during the first 15-20% of the total reaction change. Figure 1:Characteristics progress curve of an enzyme-catalysed reaction
Measurement of enzyme activity • The major factors contribute to the declines in the reaction rate: 1. Depletion of the substrate and increasing amount of product competing for the enzyme. 2. Inactivation of the enzyme, particularly if an appreciable reaction time is involved.
Measurement of enzyme activity • The indicator reaction in many coupled assays will often show a demonstrable change after the addition of the sample but before the addition of the substrate for the test enzyme. This blank reaction may be due to the presence of endogeneous substrates in the sample and its rate (B) must be measured in order to be able to calculate the activity of the test enzyme (T-B) from the total rate of reaction (T) which results from adding the substrate. Figure 2: Typical reaction trace of a coupled assay
Measurement of enzyme activity Assays for measurement reaction velocity
Spectrophotometric methods • The two forms of coenzymes (oxidized and reduced) that have different spectral characteristics. Figure 3: Adsorption spectra of NAD+ and NADH.
Immobilized enzymes Immobilized Enzymes: Enzymes physically confined or localized in a certain defined region of space with retention of their catalytic activities, and which can be used repeatedly and continuously.
Immobilized enzymes General methods of enzyme immobilization
Immobilized enzymes General methods of enzyme immobilization (cont)
Immobilized enzymes Applications of enzyme immobilization
Immobilized enzymes Table 1: Substrate assays using enzyme electrodes