Enzymes
1.17k likes | 2.36k Views
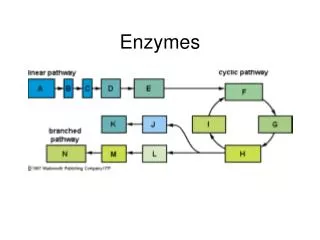
Enzymes. Enzymes. Enzymes are usually proteins which act as biological catalysts for metabolic reactions Enzymes enhance the rate at which the chemical reactions of a cell take place. They do not affect the equilibrium constant. Mechanism of Enzyme Action.

Enzymes
E N D
Presentation Transcript
Enzymes • Enzymes are usually proteins which act as biological catalysts for metabolic reactions • Enzymes enhance the rate at which the chemical reactions of a cell take place. They do not affect the equilibrium constant.
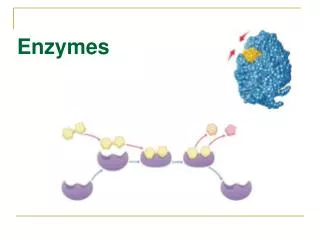
Mechanism of Enzyme Action • Enzymes interact with reactants at specific places in the enzyme structure termed the active site. • The enzyme is specific for a given substrate. • The enzyme substrate complex is then converted to product which is released.
Enzyme Substrate Complex E + S <----->ES <----------> P Thus in a closed system free enzyme availability limits the rate of the reaction in the presence of saturating amounts of substrate.
Enzyme Substrate Complex Fig. 8.16 p.160
Enzyme Kinetics • Vmax = maximum rate an enzyme reaction can occur at saturating substrate concentration. • Km = the substrate concentration which gives 1/2 Vmax
Lowering the Activation Enzymes Work by Energy of a Reaction • Even exergonic chemical reactions do not occur spontaneously but require enough energy input to raise the reactants to a “transition state”. • Transition state in between the free state of reactants and free state of products.
Enzymes and substrate interactionInduced Fit Model • The induced fit model says that the specific site for substrate is a very close fit but not exact. • When the substrate hits the active site it binds and induces a change in the protein which positions and stresses the substrate so that a reaction is most likely to occur.
Enzymes As Catalysts Enzymes work by: • Concentrating them at the active site • Poising the substrate to lower the activation energy and correctly orient them in a position most likely to allow for a reaction.
Influence of Environment on Enzyme Activity • Effect of Temperature. The Arrhenius Plot. Enzyme reactions will double in rate for every 10C increase in temperature until they reach an optimum. • Effect of pH. Most enzymes have an optimal pH between 7 and 8.
Enzyme Composition Enzymes which are made only of protein • Those whose activity is optimal with only substrate • Those which require external cofactors to be fully active: Cofactors include such things as Mg+2 or Mn+2, NAD,FAD, Zn+1, etc
Enzyme Composition Enzymes which contain organic or inorganic molecules covalently bound to proteins. • Apoenzyme describes the protein part of such an enzyme • Prosthetic group is the non protein component • Holoenzyme is the entire complex
Enzyme Composition Examples of Holoenzymes • Cytochromes. Heme = prostetic group. • Aconitase. Enzyme of the TCA cycle. FeS is the prosthetic group • Alpha Ketoglutarate dehydrogenase and pyruvate dehydrogenase. Dihydrolipoic Acid is the prosthetic group
Examples of allosterism, aspartate carbamoyltransferase and carbamoyl phosphate synthetase
Metabolism • Solute Transport • Catabolism-Breaking complex molecules into simple molecules to generate biological energy • Anabolism-Building macromolecules (lipids, protein, nucleic acids and polysaccharides) from simple molecules. Biosynthesis
Transport • Simple Diffusion: gases, water • Facilitated or passive transport: glycerol • Active transport: most solutes • Group translocation: sugars including glucose and other sugars such as fructose, mannose, maltose and sucrose.
Transport Simple Diffusion • No protein carrier molecule and therefore does not obey saturation kinetics • No biological energy requirment • Does not work against a concentration gradient • Examples: water (osmosis), gases.
Facilitated Diffusion • Requires a protein carrier thus obeys saturation kinetics • No biological energy requirement • Cannot move solute against a concentration gradient • Example : glycerol transport
Active Transport • Requires metabolic energy • Requires protein carrier and thus obeys saturation kinetics • Moves solute against a concentration gradient • Example: amino acid transport systems
Specific Active Transport Systems of E. coli Binding Protein Transport Systems • Metabolic energy source is ATP directly • Binding proteins located in periplasmic space bind substrate • Substrate carrying binding proteins lock with cytoplasmic membrane carriers which transport substrate using ATP as energy. Histidine transport is an example.
Gradient Driven Active Transport • Metabolic energy used is usually proton motive force (H+ gradient) established through respiration • No binding protein but has transmembrane carrier protein • Systems incude Antiport, Symport and uniport. Examples, sodium export, lactose transport and nitrite export
Group Translocation Phosphoenolpyruvate:sugar phosphotransferase system • Requires no net metabolic energy • Requires protein carrier as well as cytoplasmic proteins • Solute is modified during transport and therefore cannot discuss concentration gradients
Microbe of the Week • Salmonella typhi • Gram-negative, motile, mesophilic enteric bacterium • Causative agent of typhoid fever (aka “enteric fever”
Typhoid fever: the illness 7-28 days (avg. 14 days) Fever, malaise, anorexia, spots on trunk Diarrhea or constipation Delirium 75% hospitalized Fatality rate = 0.4% Recovery: 1-8 weeks
Sources • Humans are sole reservoir (does not infect animals) • Carriers may harbor the organism in their gall bladder • Contaminated food – by handlers (milk, sandwiches, meat, cake!) • or … • Contaminated water – e.g. shellfish in polluted waters • Organism survives in shellfish up to 4 days, sea water up to 9 days, for weeks in sewage • Transmission: mainly from water contaminated with human waste or human carriers
Typhoid Mary Sociological implications of infectious disease • Typhoid Mary's real name was Mary Mallon. • Irish immigrant who made her living as a cook • Mallon was the first person found to be a "healthy carrier" of typhoid fever in the United States. • She herself was not sick – but over 30% of the bacteria in her feces were S. typhi • Mallon is attributed with infecting 47 people with typhoid fever, three of whom died. • Interred on a N. Brother Island, NY for 26 years • 1907-1910 • 1915- till her death in 1938
Sociological Implications of Infectious Disease Typhoid Mary Mary Mallon (wearing glasses) photographed with bacteriologist Emma Sherman on North Brother Island in 1931 or 1932, over 15 years after she had been quarantined there permanently.
The Two General Mechanisms for Making Energy • Substrate Phosphorylation: ATP is made directly through a specific enzymatic exergonic reaction. Examples: 3 phosphoglycerate kinase and pyruvate kinase. • Respiratory driven proton translocation coupled with ATP synthesis otherwise known as the Chemiosmotic Mechanism
Substrate Phosphorylation General Concept A + B + ADP + Pi<--------->C + D + ATP Usually specific reactions of glycolysis are given as examples of these kinds of reactions
Chemiosmotic Theory Couples respiration with a proton gradient that can be used to drive ATP synthesis through the ATPase enzyme
Requiring ATP 1. Glucose + ATP------->glucose-6P + ADP (6C) Hexokinase (6C) 2. Fructose-6P + ATP----->Fructose-1,6 bisphosphate +ADP (6 carbons) phosphofructokinase (6carbons) Yielding ATP 1,3 bisphosphoglycerate+ADP----->1phosphoglycerate + ATP (3Carbons)phosphoglycerate kinase(3Carbons) Phosphoenolpyruvate + ADP-----------> Pyruvate + ATP (3Carbons)pyruvate kinase(3Carbons) Net Gain from substrate phosphorylation per glucose=2ATP ATP requiring and ATP yielding reactions in glycolysis
Specific Glycolytic Reaction forming Reduced NAD Glyceraldehyde 3 phosphate + NAD + Pi<------->1,3 bisphosphate glycerate glyceraldehyde 3 phosphate dehydrogenase