Alkyl Halides
Alkyl Halides. Nucleophilic Substitution and Elimination. Nomenclature of Alkyl Halides. Name halogen as substituent on alkane or cylcoalkane. Learn common names for some of the simple structures. e.g. chloroform, methylene chloride.

Alkyl Halides
E N D
Presentation Transcript
Alkyl Halides Nucleophilic Substitution and Elimination
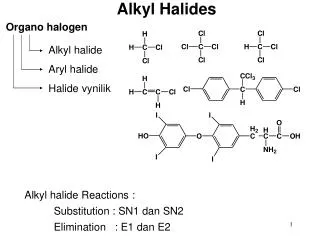
Nomenclature of Alkyl Halides • Name halogen as substituent on alkane or cylcoalkane. • Learn common names for some of the simple structures. e.g. chloroform, methylene chloride. • Note degree of substitution - name as type of C it is bonded to (i.e. 10, 20, 30). • Geminal (gem-) dihalide has two halogen atoms bonded to the same carbon. • Vicinal (vic-) dihalide has two halogens bonded to adjacent carbons. Do problem 6-1, 6-2 and 6-3 of the text.
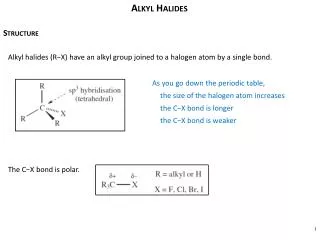
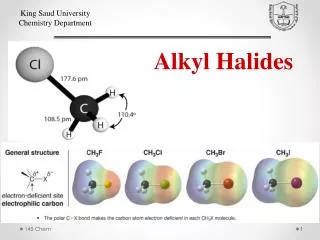
Uses and General Chracteristics • BOND DIPOLE (): + at C, - at X • all reactions based on this. • The bond dipole moments increase in the order: C—I < C—Br < C—F < C—Cl • Physical properties • generally, trends are similar to those seen in alkanes. • bp affected by London forces and dipole-dipole attractions. • Common uses: solvents, anesthetics, freons (refrigerants), pesticides.
Preparation of alkyl & allylic halides • Free radical halogenation of alkanes (Chpt 4) • You are expected to know the mechanism by which this transformation takes place.
Preparation of Alkyl Halides • Free radical halogenation of alkenes at allylic position • need to know resonance structures for intermediate & predict major/minor product See pages 235-236 of the text. Do problems 6-8 and 6-9.
Nucleophilic Substitution (SN) R—LG + Nuc: R—Nuc + LG: • Substrate • Reagent/Nucleophile (Nuc) • Leaving Group (LG) • Solvent/Reaction Conditions
1. Identify electrophilic carbon in substrate 2. Identify nucleophilic electrons in nucleophile 3. Identify leaving group in substrate
Then draw product(s) 4. Draw substrate without LG but with bond 5. Add Nuc to bond where LG used to be
Factors influencing what products are formed • Substrate/steric effects • Strength of nucleophile vs. basicity of nucleophile • Stability of leaving group • Reaction conditions • Polarity of solvent • acidic/neutral/basic
Substitution Mechanisms • Continuum of possible mechanisms • Mechanism determined primarily by substrate steric effects SN2 - methyl, 1º & unhindered 2º SN1 - 3º, hindered 2º
Bimolecular (SN2) Nucleophilic Substitution • concerted reaction; Nuc attacks, LG leaves • pentacoordinate carbon in transition state • rate depends on conc. of both reactants • Me = methyl group; Et = ethyl group
Reaction is “stereospecific” • 100 % inversion of configuration You should know how to represent this mechanism in an energy diagram!!
Factors that Affect SN2 Reaction Rates • Strength of Nucleophile: species with negative charge is a stronger nuc than an analogous neutral species (e.g. -OH > H2O; -NH2 > NH3). • Nucleophilicity increases from left to right across the periodic chart (e.g. -OH > -F). • Nucleophilicity increased down the periodic table (I- > Br- > Cl- > F-) or (-SeH > -SH > -OH). • Solvent: Polar protic solvents (e.g. ethanol, ammonia decrease nucleophilicity. Polar aprotic solvents e.g. (acetonitrile, DMSO, acetone) increase nucleophilicity.
Factors that affect SN2 reaction rates • Steric Effects: When bulky groups interfere with a rxn. because of their size, this is called steric hindrance. Steric hindrance affects nucleophilicity, not basicity. (e.g. ethoxide ion is a stronger base than t-butoxide ion). Also, alkyl halide reactivity decreases from methyl to 10 to 20 to 30. In fact, 30 alkyl halides do not react by SN2. • Leaving group: The substrate should have a good leaving group. A good leaving group should be electron withdrawing, relatively stable, and polarizable. They are weak bases. Examples are Cl-, Br-, I-, RSO3-, RSO4-, RPO4-, and neutral molecules such as water, alcohols and amines. Strong bases (OH-, RO-, H2N-) are not good leaving groups!
Unimolecular (SN1) Nucleophilic Substitution • Two-step reaction • LG leaves, then Nuc: attacks • Tricoordinate carbocation intermediate • Solvolysis (when solvent is also the nucleophile = SN1 reaction • Rate depends on substrate conc. only
Mechanism of SN1 reaction You must be able to represent this on an energy diagram!
Reaction Not StereoselectiveUnless Steric Factors Apply • Racemization - not always exactly 50/50. Carbocation can be attacked from the top or bottom face giving both enantiomers. • Steric hindrance gives attack at one side preferentially • Longer-lived carbocations give more racemization, shorter-lived give more inversion
Factors Influencing SN1 Reaction Rates • Stability of the carbocation* Allylic 3° >> 3° allylic 2° > 2° allylic 1° >> 1° > Me Carbocations are stabilized by alkyl groups (through hyperconjugation and the inductive effect) and by resonance. • Leaving group stability: the better the leaving group, the faster the reaction. • Solvent polarity: the reaction is favored in polar protic solvents. *must have neutral to acidic conditions to form carbocation
Rearrangement of Carbocations • Large difference in energy (stability) of 3° vs. 2° C+ • H- (hydride) or R- will shift (migrate) to adjacent position to form more stable carbocation. E.g. when neopentyl bromide is boiled in methanol, only rearranged product is formed.
Elimination Reactions • May proceed by a unimolecular (E1) or bimolecular (E2) mechanism. • In an alkyl halide, when a halide ion leaves with another atom or ion, the reaction is an elimination. • If the halide ion leaves with H+, the reaction is called a dehydrohalogenation.
Elimination Mechanisms • Mechanism determined primarily by substrate steric effects
E1 & SN1Competition • Always • by definition a nucleophile is Lewis Base
Carbocations generally always give both products • Relative amounts not easily predictable • Always assume formed in approximately equal amounts
E2 is Stereospecific • anti-coplanar elimination of H and LG
Product Distribution in E2 • Seytzeff Product, most substituted • major with small base, i.e., ethoxide, small LG + minor major R2C=CR2 > R2C=CHR > RHC=CHR > R2C=CH2 > RHC=CH2 Decreasing alkene stability
E2 Mechanism • Concerted, anti-coplanar, Stereospecific • strong base & good LG
Comparison of SN1 and SN2 E1 E2 • Strong bases required • Substrates: reactivity order is 3o > 2o > 1o • Solvent polarity is not so important • Rate: depends on conc. of substrate and base. • Stereochemistry: coplanar arrangement required in transition state; Saytzeff rule followed • Rearrangements: not possible • Base strength unimportant • Substrates: reactivity order is 3o > 2o > 1o • Solvent: good ionizing solvent required • Rate: depends on substrate conc. only • Stereochemistry: no particular geometry required for slow step; Saytzeff rule followed • Rearrangements: very common