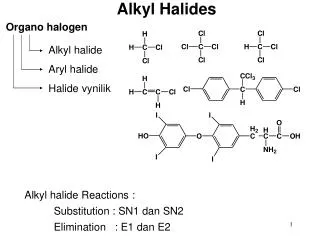
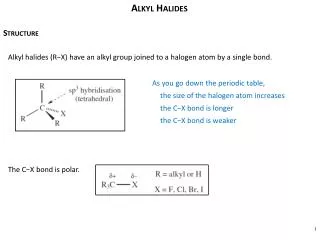
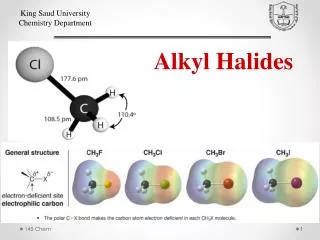
Alkyl Halides
Alkyl Halides. Name the following alkyl halides. How are alkyl halides prepared?. From alkanes Free radical halogenation From alkenes Hydrohalogenation Halogenation From alcohols. Free Radical Chlorination. Experimental Evidence Helps to Determine Mechanism.

Alkyl Halides
E N D
Presentation Transcript
How are alkyl halides prepared? • From alkanes • Free radical halogenation • From alkenes • Hydrohalogenation • Halogenation • From alcohols
Experimental Evidence Helps to Determine Mechanism • Chlorination does not occur at room temperature in the dark. • The most effective wavelength of light is blue that is strongly absorbed by Cl2 gas. • The light-initiated reaction has a high quantum yield (many molecules of product are formed from each photon of light).
Free Radical Species are Constantly Generated Throughout the ReactionPropagation
(1) Consider the free radical monochlorination of 2,2,5-trimethylhexane. Draw all of the unique products (Ignore stereoisomers). (2) Consider the free radical monochlorination of 1,4-dimethylcyclohexane. The reaction affords three different products (all C8H15Cl). Draw the three products (Ignore stereoisomers).
Conversion of Alcohols into Alkyl Halides Reactions with HX, SOCl2, PBr3
Conversion of Alcohol into a Leaving Group • Form Tosylate (p-TsCl, pyridine) • Use strong acid (H3O+) • Convert to Alkyl Halide (HX, SOCl2, PBr3)
What the **** is SN1? Unimolecular Nucleophilic Substitution
How would you prepare the following RX from the appropriate alcohols?
7.25] Draw and name the monochlorination products of the reaction of 2-methylpentane with chlorine gas. Which are chiral? 7.36] How would you prepare the ff cpds, starting from cyclopentene and any other reagents needed? • Chlorocyclopentene • Cyclopentanol • Cyclopentylmagnesium chloride • cyclopentane
Reactions of Alkyl Halides Grignard Reagents
Outline a synthetic scheme from the appropriate alkyl halide
Reactions of Alkyl Halides Substitution and Elimination
Substrate Reactivity Since the energy of the transition state is significant in determining the rate of the reaction, a primary substrate will react more rapidly than secondary (which is much more rapid than tertiary).
Bimolecular Elimination - E2Nucleophile acts as Bronsted Base