TELOMERES & TELOMERASE
TELOMERES & TELOMERASE. Prasamit Saurav Baruah MODERATOR Dr. Nalini K. Objectives :. Introduction Overview of DNA replication End replication problem Telomeres – structure & functions Telomerase – hTR & hTERT Telomere replication Regulation of Telomerase TRAP assay _ overview

TELOMERES & TELOMERASE
E N D
Presentation Transcript
TELOMERES &TELOMERASE Prasamit Saurav Baruah MODERATOR Dr. Nalini K.
Objectives : Introduction Overview of DNA replication End replication problem Telomeres – structure & functions Telomerase – hTR & hTERT Telomere replication Regulation of Telomerase TRAP assay _ overview Telomeres & cancer Conclusion References
Introduction • Telomeres , the DNA structures at the termini of linear chromosomes have been implicated in regulating the lifespan of normal human cells. • Replication of chromosome ends pose a special problem for cells . • Telomeres cant be replicated each time a cell divides and they shorten until reaching a critical length that signals replicative senescence.
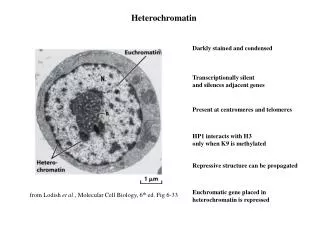
Telomere biology • Observation by HENRY MULLER , while studying Drosophila chromosomes & of BARBARA McCLINTOCK studying maize chromosomes suggested end capping of chromosomes. • MULLER coined telomere . • It’s a simple sequence of 5’-TTAGGG-3’ in humans .
Telomeres : structure • Majority of them are double helical with GC rich sequence. • The 3’ end of the DNA protrudes as a single strand overhang • Telomeres shrink at a rate of approx. 100bps per cell division from their original size of about 15 kbp.
Telomere length are the measuring stick that determines the no. of cell divisions . • Hayflick limit • Mortality stage 1 ( M1) • Mortality stage 2 ( M2)
TE L O M E R E L E N G T H Germ line cell hTERT transduced cell Stem cell Telomerase stabilization Abrogation of p53 M1 Telomerase activation M2 Crisis Senescence CELL DIVISIONS
Functions of telomeres : • Protects the chromosome from end to end fusion . • Provides a means for complete replication of chromosomes. • Contributes to the functional organization of chromosomes within the nucleus. • Participates in the regulation of gene expression. • Serves as a molecular clock that controls the replicative capacity of human cells and their entry into senescence.
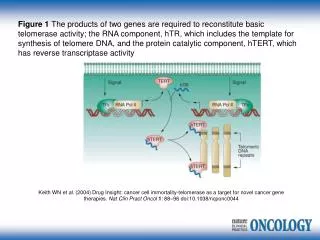
Telomerase • Its an RNA dependent DNA polymerase that synthesizes telomeric DNA sequences. • First discovered in Tetrahymena thermophila . • Telomerase activity was found to be almost absent in normal human somatic cells but present in over 90% of cancerous and in vitro immortalized cells.
Structure : • Consists of two essential components – • hTR ( HUMAN TELOMERASE) – RNA component serving as a template for telomeric synthesis. • h TERT( HUMAN TELOMERASE REVERSE TRANSCRIPTASE) - catalytic protein with Reverse Transcriptase activity.
it provides a scaffold for binding of RNP proteins • telomerase RNA feature a pseudoknot structure located close to the template sequence • 3'-half of telomerase RNA carries a box H/ACA small nucleolar RNA-like (snoRNA-like) domain
four conserved secondary structure elements, namely the 5'-terminal pseudoknot domain, the CR4–CR5 domain, the box H/ACA snoRNA-like domain and the CR7 domain . • play important roles in the function, stability, processing and intracellular trafficking of telomerase RNAs. • the box H/ACA snoRNA-like domain direct the 3'-endprocessing of precursor telomerase RNA, provide metabolic stability for the mature RNA
hTERT • Initially purified from Euplotes aediculatus as p123 containing RT’ase activity
Telomerase regulation • hTR associated proteins. • hTERT associated proteins.
Telomere associated proteins • Telomere repeat binding factor- TRF1 & TRF2. • TRF1 interacts with Tankyrase &TRF 1interacting protein 2 ( TIN2). • TRF2 interacts with hRap 1 and Mre11/ Rad 50/ Nbs 1 DNA repair complex. • Ku 70/80 heterodimer interacts with TRF2.
Protein Interacting region Function hTERT associated TEP1 aa 1–350, 601–927 Unknown P23/p90 aa 1–195 Assembly/conformation 14-3-3 aa 1004–1132 Nuclear localization hTR associated TEP1 nt 1–871 Unknown hGAR1 hTR H/ACA domain Stability, maturation, localization Dyskerin/NAP57 hTR H/ACA domain Stability, maturation, localization hNOP10 hTR H/ACA domain Unknown hNHP2 hTR H/ACA domain Stability, maturation, localization C1/C2 nt 33–147 Stability, maturation, localization La nt 1–205, 250–451 Accessibility to telomeres? A1/UP1 nt 1–208 Unknown hStau nt 64–222 Accessibility to telomeres? L22 nt 64–222 hTR processing, localization?
Telomeres are tucked into double strand repeat forming a loop , which may serve to protect the 3’ overhang. Displacement of TG rich strand internally stabilized by TRF2. Rest duplex is bound by TRF1. Structure of telomeres
Telomerase regulationcontd…. • In normal human cells telomerase activity appears to be strictly regulated during development. • Remains active in some tissues – male germ cells, activated lymphocytes & certain types of stem cell populations.
The regulation of telomerase activity occurs at – • Transcription • mRNA splicing • Maturation & modification of hTR and hTERT • Transport and subcellular localization of each component. • Assembly of telomerase.
Activation of hTERT transcription • C-Myc • oncogene induces hTERT expression and telomerase activity in normal human mammary epithelial cell and primary fibroblasts. • Sp1 • hTERT core promoter contains five GC-boxes that are binding sites for the transcription factor Sp1. Sp1 is found to co operate with c-Myc
Human papilloma virus 16E6 • in primary human keratinocytes & mammary epithelial cells. • Steroid hormones > estrogen and androgen.
NEGATIVE REGULATORS: • Mad1 > Mad / Max heterodimers inhibit hTERT promoter. • p53 > inhibits tumor formation & hTERT promoter along with E2F, p16,p21,p15. • IFN–α inhibits hTERT promoter. • Progesterone.
Cancer • Most human cancers ‘ve short telomeres and express high levels of telomerase . • Reactivation and upregulation of telomerase activity and its hTR and hTERT are associated with all cancers with few exceptions. • Studies have found shortened telomeres in many cancers, including pancreatic, bone, prostate, bladder, lung, kidney, and head and neck.
Importantly, telomerase can cooperate • with oncogenes or with inactivation of tumor suppressor genes to induce tumorigenic conversion of normal human epithelial cells and fibroblasts.
Measuring telomerase may be a new way to detect cancer. • If scientists can learn how to stop telomerase, they might be able to fight cancer by making cancer cells age and die. • Blocking telomerase could impair fertility, wound healing, and production of blood cells and immune system cells
Do telomeres play a role in other diseases? • Dyskeratosis congenita • mis-formed nails, altered skin pigmentation and hair loss. • spots on the skin, intestinal disorders, softening of the bones and learning disabilities
Telomeres and transplanted cells • In SCID, • One approach to gene therapy is to • remove cells from the patient, • transform them with the gene for the product that the patient has been unable to synthesize, • return them to the patient
this procedure used with children suffering from severe combined immunodeficiency (SCID) • The cells described by Bodnar et. al. • these cells continue to divide indefinitely as cancer
show contact inhibition as normal cells do when grown in culture. • do not grow into tumors when injected into immunodeficient mice (as cancer cells do). • still fussy about their diet — unable to grow on the simple media that supports cancer cells in culture.
Inhibition of telomerase • One of the strategies for the development of anti-cancer therapies is to inhibit telomerase activity in cancer cells. • Inhibiting telomerase activity should result in telomere shortening and therefore cause aging and death of cancer cells. • oligonucleotides, that target the template region, or active site, of telomerase. • GRN163 and GRN163L
GRN163L is a 13-mer oligonucleotide N3’ --> P5’ thio-phosphoramidate (NPS oligonucleotide) directly bind to the RNA component of telomerase at the active site of the enzyme
Telomerase inhibition with an oligonucleotide telomerase template antagonist • 3'-azido-2',3'-dideoxy-2-aminoadenosine (AZddAA) • 3'-azido-2',3'-dideoxyadenosine (AZddA), • 9-(3-azido-2,3-dideoxy-ß-D-ribofuranosyl)-2-aminopurine (AZddAP), • 3'-azido-2-chloro-2',3'-dideoxyadenosine (AZddClA)
Extension of Life-Span byIntroduction of Telomerase intoNormal Human Cells
References • Telomerase and cancer –jerry w. shay,Woodring W. Right et . al.- Human Molecular genetics – 2001,vol 10 , no. 7. • Telomere , telomerase and cancer – Scientific American 2/96 • Telomeres and telomerase – Genes and development ,1999, 13 :2353-2359 • Telomere and telomerase – Maria A.Blaso,Susan M. Gasser, - meeting review-genesdev.org • Telomere dynamics in cancer – Nature medicinés, 6,849-851:2000 • Telomere shorten during aging – nature, 345,458-460:1990
The multistep nature of cancer-Trends inGenetics,9,138-144:1993 • Harrington et.al.- A Mammalian Telomere Associated Protein – Science-275, 973-7 • Alberts et,al.- Molecular Biology Of TheCell-2002 • Watson et.al. – Molecular Biology Of the Gene – 5th edition