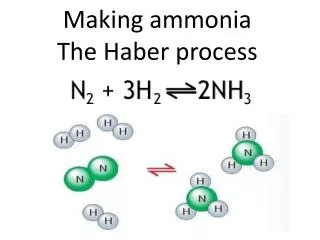
The Haber Process
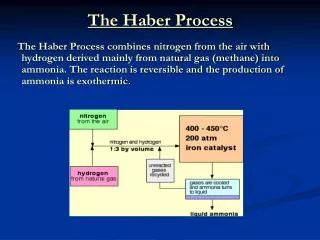
The Haber Process. How it works. Getting the nitrogen and hydrogen. They firstly extract nitrogen from the air by fractional distillation and collect this

The Haber Process
E N D
Presentation Transcript
Getting the nitrogen and hydrogen • They firstly extract nitrogen from the air by fractional distillation and collect this • They then get the hydrogen from burning hydrocarbons such as methane, with high pressured steam and a catalyst, giving hydrogen, CO and some buckminster fullerene.
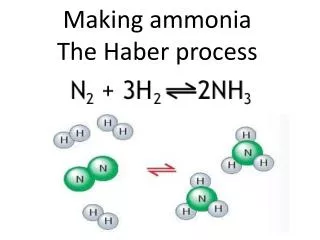
Making the ammonia • Next they pump the pressurised nitrogen and hydrogen into a chamber at 450°C with an iron catalyst • Some will turn to ammonia and it will all be cooled, with the unreacted nitrogen and hydrogen being recycled
What happens to the ammonia next? • The ammonia is cooled to a liquid and put in containers. It is then sold to farmers to fertilise their crops and get a better yield
What amount of each gas is needed? • To make ammonia in the Haber Process they use 1 part nitrogen to 3 parts hydrogen and ammonia is NH3 • This means that for it to be the most efficient they need that relative amount of hydrogen and nitrogen