HABER PROCESS
HABER PROCESS. NECESSARY FOR EVERYDAY LIFE. Contents. What? Why? Origin of Haber process Production of Ammonia now Uses(Applications) of Ammonia. WHAT IS IT?. HABER PROCESS is to produce ammonia from nitrogen and hydrogen. N 2 + 3H 2 NH 3. WHY?. AMMONIA is VERY useful in many ways.

HABER PROCESS
E N D
Presentation Transcript
HABER PROCESS NECESSARY FOR EVERYDAY LIFE
Contents • What? Why? • Origin of Haber process • Production of Ammonia now • Uses(Applications) of Ammonia
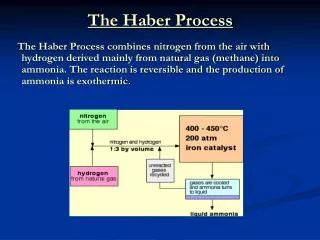
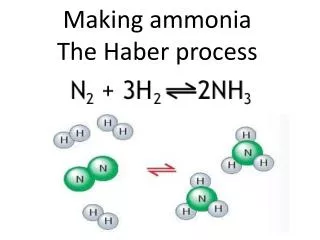
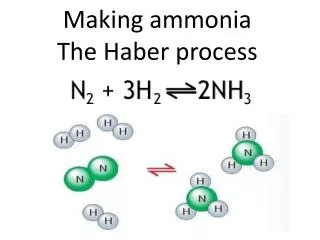
WHAT IS IT? • HABER PROCESS is to produce ammonia from nitrogen and hydrogen. • N2 + 3H2 NH3
WHY? • AMMONIA is VERY useful in many ways. • Making ammonia is cheap. • Uses of Ammonia will be explained later.
Origin • German Chemist Fritz Haber found this process at the beginning of the 20th century, right before WWI. • Before that Germany imported nitrates from South America, but the demand of Nitrates increased before the war.
Origin • During WWI, Allies blocked all the trade routes of Nitrates to Germany, but Germany was still able to produce fertilizers and explosives because of the Haber process.
How? • Haber process is done in big factories now, like…
Compressor Catalyst Condenser Gases mixed & scrubbed Liquid NH3 N2 & H2
Obtain hydrogen • Most hydrogen now is produced from “Steam-Methane Reforming”, methane is reacted with high-temperature steam(under high pressure) to form carbon monoxide and hydrogen. CH4 + H2O (+heat) → CO + 3H2
Obtain hydrogen • After that the carbon monoxide is combined with steam (+catalyst) and carbon dioxide and hydrogen. CO + H2O → CO2 + H2 (+heat)
Obtaining hydrogen • Electrolysis is also used to obtain hydrogen, but greenhouse gas is produced from burning fossil fuels to make electricity, so this method is less common compared to “Steam-Methane Reforming”.
Obtaining Nitrogen • Nitrogen is obtained from the air. (of course) • Air is cooled to ≈-200oC, then fractionally distilled.
Uses of Ammonia • Fertilizers 75% • Nitric Acid 10% • Nylon 5% • Others 10%
Nylon • Polymerization.
Nitric Acid • Ammonia is reacted with Oxygen (catalyst platinum-rhodium gauze) to produce nitrogen dioxide and water. • The nitrogen dioxide is then dissolved in water (with air)
Nitric acid eqns • 4NH3 + 7O2 4NO2 + 6H2O • 4NO2 + 2H2O + O2 4HNO3
Fertilizers • To make fertilizers, nitrates are required. • There are two types of fertilizer, straight-N & NPK fertilizers.
Fertilizers • Example of Straight N fertilizer is Ammonium Nitrate. • Ammonia + Nitric Acid => Ammonium Nitrate
Importance? today • Some people say Haber process and ammonia is the most important invention or the past few hundred years • But others say that contact process is the most important.
According to Mr. Ed (last year’s chemistry teacher)Sulfuric acid is the “KING OF CHEMICALS” • So Haber process comes in 2nd
Sources • Keroballhttp://www.mana.co.jp/kerokuma.html • Ammoniahttp://www.dicts.info/picture-dictionary.php?group=&w=ammonia&search=Search&language=english • Fertilizer http://www.hydrangeashydrangeas.com/fertilize.html • HNO3 http://commons.wikimedia.org/wiki/File:Nitric-acid-3D-balls-B.png • History http://www.easychem.com.au/monitoring-and-management/maximising-production/history-of-the-haber-process • Electrolysis http://www1.eere.energy.gov/hydrogenandfuelcells/production/electro_processes.html • Gas reforming http://www1.eere.energy.gov/hydrogenandfuelcells/production/natural_gas.html • Background pictures from euro truck simulator 2 • And Cambridge IGCSE Chemistry Course book