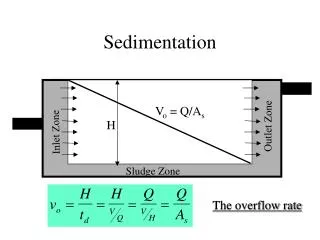
2. sedimentation
2. sedimentation. ULTRAFILTRATION. Suspended materials and macromolecules can be separated from a waste stream using a membrane and pressure differential, called Ultrafitration . The difference between Ultrafitration & reverse osmosis :

2. sedimentation
E N D
Presentation Transcript
ULTRAFILTRATION Suspended materials and macromolecules can be separated from a waste stream using a membrane and pressure differential, called Ultrafitration. The difference between Ultrafitration& reverse osmosis : This method uses a lower pressure differential than reverse osmosis and doesn't rely on overcoming osmotic effects.
How ultrafiltration works? Water and some dissolved low molecular weight materials pass through the membrane under an applied hydrostatic pressure. Emulsified oil droplets and suspended particles are retained, concentrated, and removed continuously as a fluid concentrate. The pore structure of the membrane acts as a filter, passing small solutes such as salts, while retaining larger emulsified and suspended matter. The pores of ultrafiltration membranes are much smaller than the particles rejected, and particles cannot enter the membrane structure. As a result, the pores cannot become plugged.
Recovery of paint from water soluble coat bases (primers) applied by the wet electrodeposition process (electrocoating) in auto and appliance factories. • Recovery of proteins in cheese whey (a waste from cheese processing) for dairy applications. • The concentration of waste-oil emulsions from machine shops, which are produced in association with cooling, lubricating, machining, and heavy metal rolling operations. • Water pollution analysis to concentrate organisms from water samples. IMPORTANT APPLICATIONS
In the case of microfiltration, a more porous membrane is used than in the other membrane separation technologies, thus yielding a relatively higher flux. It is mainly useful in removing turbid causing materials and can replace conventional granular filtration processes. • The most significant design parameters are: • Transmembrane pressure • Size and geometry of modules • Tangential velocity • Recirculation factor MICROFILTRATION & NANOFILTRATION
The clean water flux across a membrane without any material being deposited follows Darcy’s Law The net pressure differential across a membrane, taking into consideration the osmotic pressure is given by (ΔP - Δ II) , and hence, the expression for the permeate flux is:
The resistance due to the deposit on the surface is given by the following relationship:
Microfiltration membranes might be used to achieve very low turbidity effluents with very little variance in treated water quality. Because bacteria and many other microorganisms are also removed, such membrane disinfection might avoid the need for chlorine and subsequent dechlorination.
to enhance membrane performance a. Metal salts of iron or aluminum may also be added. b. Coagulation of colloidal materials may also increase the effective size of particles applied to membranes and increase permeate flux by 1) reducing foulant penetration into membrane pores, 2) forming a more porous cake on the membrane surface, 3) decreasing the accumulation of materials on the membrane due to particle size effects of particle transport. 4) improving the backflushing characteristics of the membrane.