Phase Change
Frozen Fire Andit Macarine Echin Paloma Andaluz Yamilo Bonono Ruperez Cabag Lagura Sulapas Espinosa Lumogdang. Phase Change. Introduction.

Phase Change
E N D
Presentation Transcript
Frozen Fire AnditMacarine EchinPaloma AndaluzYamilo BononoRuperez CabagLagura Sulapas Espinosa Lumogdang Phase Change
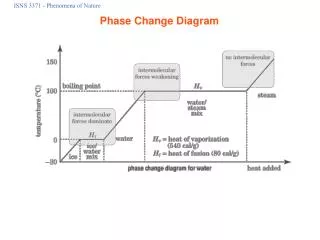
Introduction Another change that may occur when heat is added to or taken out from an object is phase change. For example, you know that water can change from solid (ice) to liquid (water) or from liquid to gas (steam).
Materials • Crushed Ice • Glass container/beaker • Timer • Stirring rod • Thermometer • Electric Burner • Wire gauze • Weighing scale
Procedures • Weigh the ice/ice cubes. Record its mass as m1. Mass of ice (m1): 423 g • Measure the temperature of the ice and assign it as the initial temperature T1. Temperature (T1): 0 degree Celsius. • Stir the contents of the container, then measure the temperature of the contents every 2 minutes after all the ice has completely melted. Record the temperature in Table 1.
Weigh the liquid contents of the beaker and record its mass as m2 and its temperature as T2. M2 = 295 g T2 = 7 degrees Celsius • Continue taking the temperature of the liquid water until the temperature starts to increase.Record the time it takes for the liquid water to increase its temperature from T2. Time taken for the temperature to start rising from T2: 1 minute • Heat the contents of the beaker. Get the temperature reading every 2 minutes until the water boils and record it in Table 2.
Continue boiling for the next 3-5 minutes and record the temperature as T3. T3 = 99 degrees Celsius
Questions • What heppens to the temperature of water while changing from ice to liquid to water? The temperature increases. • Why does ice inside the container melt after some time? Because of the factors that affects the melting of the ice, like the stirring that produces friction and gives off heat which makes melting possible and much faster.
What similarities and differences have you noticed between your graphs for table 1 and table 2?
How much heat was used to change the ice to steam? Qtotal = mcice (Tm-Ti) + mHf + mcwater (Tv-Tm) + mHv + mcsteam (Tf-Tv) = 0.423 kg (0.5 kcal/kg degree Celsius) (0 degree Celsius – 1 degree Celsius) + 0.423 kg (80 kcal/kg) + 0.295 kg ( 1 kcal/kg degree Celsius) (100 dgrees Celsius – 0 degree Celsius) + 0.295 kg (0.48 kcal/kg) (99 degrees Celsius – 100 degrees Celsius) = 63.27 kcal
Generalization An ice cube placed in a container and settled in expected to meet and when it was stirred the faster is the melting process. As the ice turns into liquid, the temperature was observed to increase. Healing the melted ice using the burner affects the temperature and evaporation occurs then.