Phase Change
Phase Change. Dec 3 rd. A Change of State. Matter can change from one state to another The attraction between molecules and their rate of movement are two factors that determine the state of matter A substance gains or loses heat when it changes state

Phase Change
E N D
Presentation Transcript
Phase Change Dec 3rd
A Change of State • Matter can change from one state to another • The attraction between molecules and their rate of movement are two factors that determine the state of matter • A substance gains or loses heat when it changes state • Changes in state can also occur because of an increase or decrease in pressure
Order of Change • When heat is applied to a substance, its change in state typically goes from solid to liquid to gas. • There are some exceptions where the material will go directly from a solid to a gas. • When a substance is cooled, its change in state typically goes from gas to liquid to solid. • There are some exceptions where the material will go directly from a gas to a solid.
Five Phase Changes The five state changes are: • Melting • Freezing • Evaporation • Condensation • Sublimation
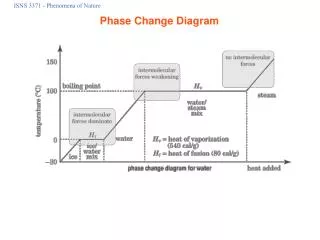
Melting (solid to liquid) • Melting occurs when a substance changes from its solid state to its liquid state and the temperature at which this happens is called the melting point. • When a solid is heated, its temperature rises until it reaches it melting point. Any additional heat added to the material will not raise the temperature until all of the material is melted • Thus, if you heat some ice, its temperature will rise until it reaches 0oC. Then the ice will stay at that temperature until all the ice is melted. The heat energy is used to melt the ice and not to raise the temperature. After the ice is melted, the temperature of the water will continue to rise as more heat is applied.
Freezing (liquid to solid) • Freezing occurs when a substance changes from its liquid state into its solid state. • The temperature at which a specific liquid becomes a solid is called the freezing point. • When cooling a liquid, its temperature will drop until it reaches the freezing point. Any additional cooling or heat loss will not lower the temperature until all of the liquid is solidified. Then the temperature of the solid can continue to decrease.
Evaporation (liquid to gas) • Evaporation is a process by which a substance changes from its liquid state to its gas state by random particle movement. • When a liquid is heated, its temperature rises until it reaches it boiling point. The temperature will then remain at that point until all of the liquid is boiled away. • For example, the temperature of a pot of water will increase until it reaches 100oC. It will stay there until all the water is boiled away. The temperature of the steam can then be increased.
Condensation (gas to liquid) • Condensation is the formation of liquid drops from water vapor. • It is the process which creates clouds, and so is necessary for rain and snow formation as well.
Sublimation (solid to gas (skipping the liquid phase)) • Sublimation happens when a frozen substance changes to a gaseous form. • The most common example of sublimation is that of dry ice, which is the common name of frozen CO2. When dry ice is exposed to the air it begins to sublimate, or change to vapor, before your very eyes. This happens to dry ice because at room temperature the frozen gas would rather be a gas than frozen solid.