PERIODIC TABLE OF ELEMENTS NOTES
PERIODIC TABLE OF ELEMENTS NOTES. Atoms vs. Ions. Atoms = same number P + & E - Ex: (+3) + (-3) = 0 charge is ZERO Ions = same # P + , …but more or less E - Ex: (+3) + (-4) = -1 Ex: (+3) + (-2) = +1 Charge is (+ or -).

PERIODIC TABLE OF ELEMENTS NOTES
E N D
Presentation Transcript
Atoms vs. Ions • Atoms = same number P+ & E- • Ex: (+3) + (-3) = 0 • charge is ZERO • Ions = same # P+, …but more or less E- • Ex: (+3) + (-4) = -1 • Ex: (+3) + (-2) = +1 • Charge is (+ or -) *sodium ion laser is used to determine the temperature in the mesosphere and the lower thermosphere
Quick Electron Lesson • Electrons are found in the energy levels. • Electrons affect the properties of elements • Remember atoms have a ZERO CHARGE • If an atom has a charge ONLY affects ELECTRONS • # P+ ≠ #E- • If Positive (+) charge, TAKE AWAY electrons • If Negative (-) charge, ADD ON electrons
Ions: What and Why? • An Ion is an atom with a + or -charge • Cation = ion with + charge • Anion = ion with - charge • Goal: fill outermost shell (Octet Rule) • Ex. Na = 11 electrons (7 more until full shell) • Na+1 = 10 electrons (outermost shell full) • Valence Electrons (outermost shell electrons) are used for bonding
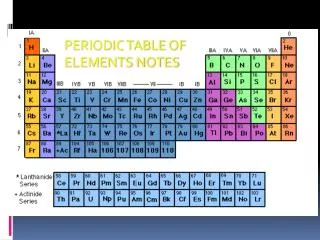
How to organize elements? • Early scientists used the properties of elements to sort them into groups. • Elements are arranged in order of increasing atomic number.
How It Works • Rows (across) = PERIODS • Goes in order of ATOMIC NUMBER • Determines # energy levels • Elements within a period = PROPERTIES CHANGE Periodic law: the pattern of properties within a period repeats as you move from one period to the next
How It Works • Columns (up and down) = GROUPS or FAMILIES • Determines the # valence electrons • Elements within a group = SIMILAR PROPERTIES
3 Broad Classes of Elements Metals Metalloids Nonmetals “Staircase”
3 Broad Classes of Elements • Metals: • Good Conductors • High Luster or Sheen (Shiny) • Ductile • Malleable • Nonmetals: (Opposite of metals) • Metalloids: Act as a metal or nonmetal
Metallic Trend • Most metallic elements are Cesium (Cr) and Francium (Fr) In your journal, outline the periodic table and include the trends.
Main Groups • Alkali Metals • Alkaline Metals • Transition Metals • Metalloids • Nonmetals • Halogens • Noble Gasses • Lanthanides • Actinides
Group 1: Alkali Metals“The Gentlemen” • Except Hydrogen • Traits: • 1 valence electron • Lose valence electron +1 charge • VERY REACTIVE • (Almost) always bonds with halogens
Group 7: Halogens “The Ladies” • Common Salts: • Chlorine (Cl) • Bromine (Br) • Iodine (I) • Traits: • 7 valence electrons • Gains 1 valence electrons -1 charge • VERY REACTIVE • Almost awalysbonds with Alkali gentlemen
Why They Make Such a Cute Couple…Reason for Bonding Goal: To settle down Meet the OCTET RULE Valence electrons = bonding Alkali Gentlemen= 1 valence electron Halogen Ladies= 7 valence electrons
In Addition…Charge • No Charge = No Drama! • Alkali (Sodium) = +1 charge • Halogens (Chlorine) = -1 charge • NaCl = (+1) + (-1) = 0
Find a Partner Who… • What charge do alkali metals have? Halogens? • What are valence electrons? • Why do alkali and halogens go so well together (two reasons)?
Group 2: Alkaline Earth Metals “Players” Players: • Traits: • 2 valence electrons • Loses 2 valence electrons +2 charge • (Almost) always bonds with TWO halogensistas OR 1 Group 6 nonmetal
Group 8: Noble Gases“The Snobs” The Snobs: • Traits: • 8 valence electrons (Octet Rule) • NO CHARGE (no drama) • NOT REACTIVE • Bonds with NO ONE • Too good for anyone else.
In between Groups:Transition Metals • Scandium to Zinc • Zinc to Mercury • Found in middle of table • Ones in each column = similar properties • Different charges • Some are synthetic • Inner Transition Metals • Lanthanides and Actinides • Rare Earth Elements
Elbow Partner • What do metalloids separate (what are the found between)? • Why are the noble gasses “snobs?” • Describe the traits of alkaline earth metals (charge, number of valence electrons).