The Atom Hypothesis
The Atom Hypothesis. As long ago as 60 BC, Lucretius reasoned that all matter must be composed some atoms – smallest indivisible units that have a given character in chemical and physical properties.

The Atom Hypothesis
E N D
Presentation Transcript
The Atom Hypothesis As long ago as 60 BC, Lucretius reasoned that all matter must be composed some atoms – smallest indivisible units that have a given character in chemical and physical properties. With the advent of chemistry and the discovery of ever-more elements, in 1869 Mendeleev realized that these elements could be ordered in a periodic table according to increasing mass and recurring chemical properties. This recursion was a clue that all atoms are made of a few more-fundamental building blocks. The building blocks are arranged in different combinations to make the various elements.
Mendeleev’s Original Periodic Table, 1869 ``I saw in a dream a table where all the elements fell into place as required. Awakening, I immediately wrote it down on a piece of paper. Only in one place did a correction later seem necessary."
Mendeleev’s Postulates • The elements, if arranged according to their atomic weights, exhibit an apparent periodicity of properties. • Elements which are similar as regards their chemical properties have atomic weights which are either of nearly the same value (eg. Pt, Ir, Os) or which increase regularly (eg. K, Ru, Cs). • The arrangement of the elements, or of groups of elements in the order of their atomic weights, corresponds to their so-called valencies, as well as, to some extent, to their distinctive chemical properties; as is apparent among other series in that of Li, Be, Ba, C, N, O, and Sn. • The elements which are the most widely diffused have small atomic weights. • The magnitude of the atomic weight determines the character of the element, just as the magnitude of the molecule determines the character of a compound body. • We must expect the discovery of many as yet unknown elements-for example, elements analogous to aluminum and silicon- whose atomic weight would be between 65 and 75. • The atomic weight of an element may sometimes be amended by a knowledge of those of its contiguous elements. Thus the atomic weight of tellurium must lie between 123 and 126, and cannot be 128. • Certain characteristic properties of elements can be foretold from their atomic weights. (18)
The Periodic Table Today = predicted by Mendeleev, discovered later
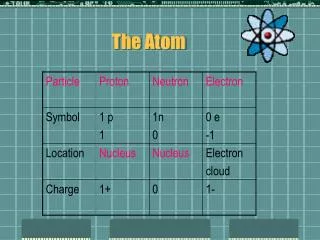
The Constituents of the Atom From stoichiometry of chemical reactions, it was known that elements combine only in fixed integer ratios their atoms must form bonds to one another, and the character of the bonds must be an inate property of each element. The size of an atom (~1 Å) was obtained from electrochemistry. The electron was discovered, from radioactivity ( rays) and from emission in cathodes ray tubes. Its charge e and mass m were measured. In electrochemical reactions, the number of electronic charges needed to react a molecular weight of a substance was measured Avogadro’s number. But the electron mass m was tiny compared to that of an atom. Conclusion: there must be positively charged particles (nucleons) within each atom, in equal number to the electrons so that the atom is neutral. The positive particles must be much heavier than the electron. The particle of radioactivity is the positive nucleus of a helium atom.
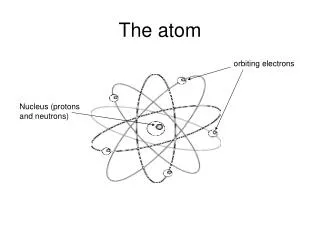
What is the structure of the nucleons and electrons in an atom? • Two possible pictures emerged: • The nuclear atom: • positive nucleus surrounded • by “solar system” of electrons • The “plum pudding” atom: • uniform distribution of • nucleons and electrons • Which picture is right?
Rutherford scattering In 1911, Ernest Rutherford set out to probe the charge distribution within the atom by scattering particles from a thin foil of gold. Plum pudding: charge distribution is everywhere locally balanced – little force, little scattering. Nuclear atom: when happens to make a near-head-on collision, it can penetrate within electron cloud – strong force, large-angle scattering.
There were troubles with either picture! Nuclear atom: What keeps the + charged nucleons bound in a small nucleus, when their Coulomb repulsion pushes them apart?? Plum pudding: How can the atoms of a substance retain their identity when the atoms are pushed together in a solid? Why don’t they just coalesce into a larger atom of another substance??
Rutherford scattering Rutherford set out to measure the distribution of scattering angle for particles, produced by radioactive decay from Polonium, scattered as they pass through a thin gold foil. Rutherford’s students observed the scattered’s on a phosphor screen (PbS film on glass, similar to the screen of a television):
distance of closest approach kinetic energy of incident : T = ½ mv2 ~ 4 MeV Potential energy when (z=2) is a distance r from nucleus: In a head-on collision, would come to a stop at a distance d where T = U:
Let’s calculate the scattering angle for an impact parameter d~2dmin: We will simplify the analysis by using the impulse approximation. Let the pass by the atom on a straight line path (no scattering). Calculate the impulse (change in momentum) from the Coulomb force acting along the trajectory. Then apply the impulse to obtain the scattering angle.
Analyze the impulse delivered by the Coulomb force separately for the directions parallel and perpendicular to the incident direction. By symmetry, the parallel impulse delivered before the reaches the atom exactly cancels the parallel impulse delivered after it has past the atom: p‖= 0. For the perpendicular impulse, calculate the perpendicular component of the Coulomb force, acting at a distance x along the path of the :
We can now calculate the scattering angle: The can only scatter at large angle if the positive charges in the atom are concentrated on a small distance scale dmin. Rutherford’s confirmation of the nuclear model of the atom raised another, deeper question: what holds the positive nucleons within such a small nucleus? Answer: the strong interaction – an immensely strong, short-range interaction that acts only within the size of the nucleus.