THE ATOM
THE ATOM. Unit 5 Chemistry Langley. ATOMIC TIMELINE. THE FIRST ATOMIC SCIENTIST. DEMOCRITUS (400 BC) First scientist to hypothesize that all matter was made of small particles Named the small particles atoms Atoms in Greek means indivisible

THE ATOM
E N D
Presentation Transcript
THE ATOM Unit 5 Chemistry Langley
THE FIRST ATOMIC SCIENTIST • DEMOCRITUS (400 BC) • First scientist to hypothesize that all matter was made of small particles • Named the small particles atoms • Atoms in Greek means indivisible • Democritus thought these particles could not be broken down into smaller units, indivisible and indestructible • Only theory, did not have “hard” evidence • His ideas were challenged by Aristotle and Plato • Aristotle (360 BC) argued that atoms did not exist
FOUNDATIONS • LAW OF CONVERSATION OF MASS • Antoinne Lavoisier clearly formulated it in 1789 • Mass is neither created nor destroyed • LAW OF DEFINITE PROPORTIONS • Joseph Proust • Specific substances always combine in the same ratio; i.e. a chemical compound always contains the same proportion of elements by mass. (Water is always 2 hydrogen and 1 oxygen) • Experiments conducted between 1797 and 1804 • What John Dalton used beginning in 1803 to formulate the basis of his atomic theory
FIRST ATOMIC THEORY • JOHN DALTON (1808) • English scientist who investigated Democritus claim of atoms • Dalton created the first “accepted” Atomic Theory by using experimental methods • All matter is made up of small particles called atoms • Atoms of a given element are identical in size and mass • Atoms of different elements can physically mix together or chemically combine to form compounds • In reactions, atoms combine, separate, or rearrange • Atoms cannot be divided, created, or destroyed
MODERN ATOMIC THEORY • 2 KEY CHANGES TO DALTON’S THEORY • Atoms are divisible into smaller particles • Given elements can have atoms of different masses
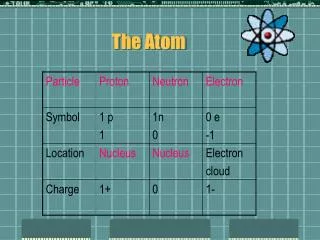
PARTICLES IN THE ATOM • DISCOVERY OF THE ELECTRON • J.J. Thomson (1897) • Created an experiment to test sending electric currents through gases at low pressure • Cathode Ray Experiment • One end, anode, became positively charged. • Other end, cathode, became negatively charged. • Once charged, a glowing beam appeared that traveled from the cathode to the anode (cathode ray). • Since the atoms are suppose to be neutral, then why would they atoms be attracted to the positive plate? • Hypothesized that there was some negative particle in the atom that was attracted to the positive plate • Named the negative particle a corpuscle
PARTICLES IN THE ATOM • DISCOVERY OF THE ELECTRON • J.J. Thomson • With the discovery of the electron, developed the Plum Pudding Model • Pictured plum like negatively charged electrons embedded in a sphere of positively charged pudding like “goo”
PARTICLES IN THE ATOM • CHARGE AND MASS OF THE ELECTRON • Robert Millikan conducting the infamous Oil Drop Experiment in 1909 (and again in 1913 due to infighting with another physicist) was able to calculate the specific mass of a single electron and prove that it does in fact have a negative electric charge • Mass of an electron is 9.109 x 10-28 kg
PARTICLES IN THE ATOM • DISCOVERY OF THE PROTON • Eugen Goldstein (1886) • Observed a cathode-ray tube and found that rays could also travel in a tube toward the cathode (negative end) • Since opposite attracts, there had to be a positive particle in the atom, called these things canal rays • Positive particles are protons and have a mass about 1840 times larger than an electron • (Sometimes because his colleagues did not agree with him, he is often not given credit with discovering the proton)
PARTICLES IN THE ATOM • DISCOVERY OF THE NEUTRON • James Chadwick (1932) • Found high energy particles with no charge and roughly the same mass as a proton (neutron is slightly larger) • Neutron has no charge
THE CENTER OF THE ATOM • THE NUCLEUS • Ernest Rutherford (1911) • Gold Foil Experiment • A piece of gold foil was bombarded with alpha particles (positively charged) • Assumed the positively charged and electrons were evenly distributed throughout the gold (Plum Pudding Model) • Of 8,000 alpha particles fired, 7,999 “stuck” to the foil (because opposite attract and the + alpha particles were attracted to the – electrons in the Au atoms • 1 in 8,000 particles bounced back. These alpha particles must have found the protons in the Au atoms
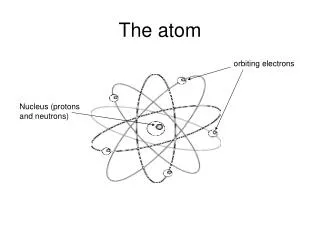
THE CENTER OF THE ATOM • THE NUCLEUS • Ernest Rutherford • Gold Foil Experiment showed only a small amount of space occupied by positive particles • Concluded that almost all the mass and positive charge of an atom is located in the center (the nucleus) and the electrons are just scattered around and orbit the nucleus like the planets orbit the sun • Later found that nucleus contains protons (positive particles) and neutrons (particles with no charge)
FORCES IN THE NUCLEUS • Nucleus held together by short range nuclear forces • Short range forces include: • Proton-Proton • Proton-Neutron • Neutron-Neutron
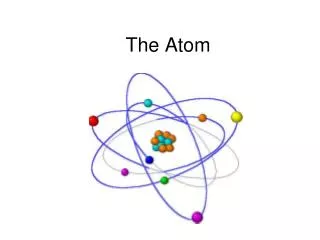
ACCEPTED THEORY TODAY • Based on Wave Mechanics • Center of the atom is called the nucleus • Contains the protons and the neutrons • Electrons “float” in the electron cloud • Electron cloud broken up into energy levels • Electrons’ distance from the nucleus depends on their energy • All matter is made up of atoms • Atoms from different elements combine to form compounds in chemical reactions; can also physically combine • Atoms rearrange, separate, combine in chemical reactions
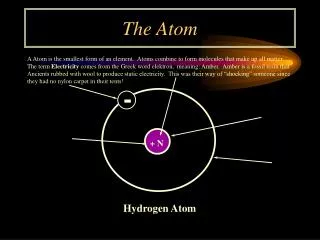
IDENTIFYING ATOMS • Atomic NumberNumber of protons in the nucleus of an atom of that element • Periodic table is arranged according to increasing atomic number • The atomic number identifies the element • Because atoms are electrically neutrally, the number of protons equals the numbers of electrons
IDENTIFYING ATOMS • Mass NumberTotal number of protons and neutrons • IS NOT ON THE PERIODIC TABLE!!!!!!!! • Atoms of the same element do NOT have to have the same number of neutrons (isotopes) • Isotope: atoms of the same element that have different masses due to different numbers of neutrons • Number of neutrons = mass # - atomic # • Example: Carbon with a mass number of 15
IDENTIFYING ATOMS • Average Atomic Mass • Since not all atoms of the same element have the same mass (because of the existence of isotopes), the mass on the periodic table (the decimal number) is the average atomic mass • This mass is given in atomic mass units (amu) • To find the average atomic mass: identify all known isotopes of an element and record their masses and determine the average
IDENTIFYING ATOMS • Average Atomic Mass • Examples: • Hydrogen • Formula: (%abudance/100)(mass)+(%abundance/100)(mass) • Neon-20 has a mass of 19.992 amu and Neo-22 has a mass of 21.991 amu. In an average sample of 100 Neon atoms, 90 will be Neon-20 and 10 will be Neon-22. Calculate the average atomic mass.
NOMENCLATURE Mass # 17 o 8 Chemical Symbol O - 17 Atomic # Mass #
MOLE CONVERSIONS WITH COMPOUNDS • MOLE REVIEW • One mole of ANY element is equal to 6.02x1023 atoms of that element • The mass (in grams) of one mole of any element is found on the periodic table underneath the symbol • Example: 45 grams of Na = ? Moles • Example: 7.8x1045 atoms of K = ? g of K
MOLE CONVERSIONS WITH COMPOUNDS • 56 grams of Al2O3 = ? Moles of Al2O3 • Molar Mass of a Compound • Determine how many of each element is present in the compound • Multiply the mass of the element times the number of that element present • Add the masses together • Convert using the fence method • 45 moles of H2O = ? of H2O • 9.4x1035 atoms of AmO = ? g of AmO
NUCLEAR RADIATION • DISCOVERY • Henri Becquerel • Uranium salts and photographic plates • Conducted experiment twice, once on a sunny day and once on a cloudy day • Marie Curie • Named the process by which particles give off rays radioactivity • Spent entire scientific career working on advancements within nuclear radiation field • Won two Nobel Prizes for work; one was shared with husband and Becquerel, one was independently wond
RADIATION DECAY • RADIOACTIVE DECAY • When a nucleus spontaneously disintegrates into a lighter, more stable element • When this happens, x rays and radiation are given off • NUCLEAR STABILITY • In nuclear reactions, the nuclei of unstable isotopes (radioisotopes) gain stability by undergoing changes. These changes will continue until stability is reached.
RADIATION DECAY • 6 TYPES OF RADIOACTIVE DECAY • Alpha Decay • Beta Decay • Positron Decay • Gamma Decay • Proton • Neutron • The neutron-to-proton ration determines the type of decay that occurs
TYPES OF RADIATION (NUCLEAR REACTIONS) • 3 MAIN TYPES • Alpha Radiation • Consists of a helium nuclei (alpha particle) emitted from a radioactive source; alpha particle emitted which contains two protons, two neutrons and has a double positive charge • Beta Radiation • An electron resulting from the breaking apart of a neutron in an atom; the neutron breaks apart into a proton. All that remains is a nucleus; the electron is released (beta particle) • Gamma Radiation • High-energy photon; electromagnetic. Nuclei often emit gamma rays along with alpha or beta particles during radioactive decay; (gamma ray-particle) no mass or electrical charge
NUCLEAR REACTIONS • EXAMPLES • 32 __ 28 P __ + Al 15 __ 13 • 14 ___ 14 C ___ + N 6 ___ 7 • 176 1 ___ Ra + n ___ 88 0 ___
NUCLEAR REACTIONS • TRANSMUTATION • The conversion of an atom of one element to an atom of another element (via changing the number of protons in the element) • Two ways it occurs • Radioactive decay • Particles bombard the nucleus • Allows chemists to produce elements that do not occur naturally • These elements have atomic numbers greater than 92 • Earliest transmutation took place in 1919 by Rutherford; took Nitrogen-14 and formed an unstable isotope of Fluorine (Fluorine-18); it was this experiment that led to the discovery of the proton
NUCLEAR STABILITY • NUCLEAR STABILITY • Helium has 2 protons, 2 neutrons, 2 electrons • Atoms only made of protons, neutrons, electrons • To find the mass of one atom Helium, add up masses of the 3 particles • Proton = 1.007 amu • Neutron = 1.009 amu • Electron = 0.001 amu • 2(1.007) + 2(1.009) + 2(0.001) = 4.033 amu • If you put that same atom on the scale, the mass is only 4.022 amuMass Defect
NUCLEAR STABILITY • MASS DEFECT • Difference between adding up the masses of all the particles in an atom and the mass of the actual atom • The EINSTEIN CONNECTION • E = mc2 • Mass moving at high speed can be converted to energy • Mass defect comes because some of the mass is changed into energy • NUCLEAR BINING ENERGY • Energy released when nucleus is formed • The higher the energy, the more stable the atom
NUCLEAR FISSION • Splitting of a nucleus into smaller fragments • Uranium-235 and plutonium-239 • Started with neutron bombardment • Continued via chain reaction • Neutrons produced react with other fissionable atoms, producing more neutrons which react with still more fissionable atoms • Continues until nucleus stability is reached • Control fission in a nuclear reaction by neutron moderation and nuclear absorption • Nuclear Power Plants • High nuclear waste • Atomic bomb
NUCLEAR FUSION • Nuclei combine to produce a nucleus of greater mass • Produces more energy than fission reactions • Sun • High temperatures required and maintained • Low waste • H bomb
NUCLEAR REACTIONS • HALF LIFE • The time required for one-half of the nuclei of a radioisotope sample to decay to products • After each half-life, half of the existing radioactive atoms have decayed into atoms of a new element
NUCLEAR REACTIONS • HALF LIFE • Example 1: If Polonium-32 has a half life of 14.3 days and you start with 4.0 mg, how many mg with you have after 57.2 days? Step 1: How many half lives with the atom go through? (time you wait)/(half life length) = 57.2/14.3 = 4.00 Step 2: Amount left = starting amount * (1/2)#half lives Amount left = (4.0)*(1/2)4.00 = ?????
NUCLEAR REACTIONS • HALF LIFE • Example 2: The half life of radon-222 is 3.824 days. If you wait for 15 days and find 50 grams of radon-222, how much did you start with?