Amines
Amines. Introduction. Amines are formed by replacing one or more hydrogen atoms of ammonia (NH 3 ) with alkyl groups. In nature , they occur among proteins, vitamins, alkaloids and hormones. Synthetic examples include polymers, dyestuffs and drugs .

Amines
E N D
Presentation Transcript
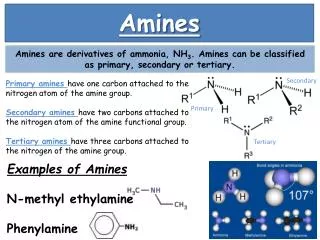
Introduction • Amines are formed by replacing one or more hydrogen atoms of ammonia (NH3) with alkyl groups. • In nature, they occur among proteins, vitamins, alkaloids and hormones. Synthetic examples include polymers, dyestuffs and drugs. • Two biologically active compounds, namely adrenaline and ephedrine, both containing secondary amino group, are used to increase blood pressure.
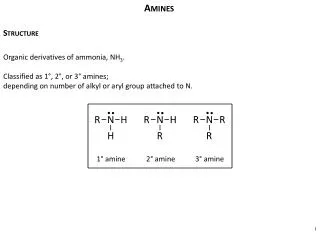
N H3C CH3 H3C Structure of Amines Nitrogen orbitals in amines are sp3 hybridized also the geometry of amines is pyramidal. Every of the three sp3 hybridized orbitals of nitrogen are related with orbitals of hydrogen or carbon depending ahead the composition of the amines
Aniline It is one of the most important of all the amines.
MANUFACTURING PROCESS 1 By Reduction of Nitrobenzene CLICK CLICK 2. By Reduction from Nitro Alkanes CLICK 3. By Reduction from Acid amide CLICK 4. By Hofmann degradation method 5. From Acid Nitrile CLICK CLICK 6. From Halo Alkane
Tin and Hydrochloric Acid Nitro Benzene Reduction with Iron scrap with Hydrochloric acid is preffered because FeCl2 formed in reaction get hydrolysed and release HCl hence small amount of HCl is required to initiate the reaction BACK
From Nitro Alkanes Other reducing agents Sn/Conc. HCl, H2/Ni BACK
Ammonolysis of alkyl halides • The process of cleavage of the C–X bond from an alkyl or benzyl halides by ammonia molecule is known as ammonolysis. The order of reactivity of halides with amines is RI > RBr >RCl.
From Acid Amide See number of carbon atoms remains same after reduction BACK
From Hoffman degradation method See the number of carbon atom get reduced by one BACK
From acid nitrile Other reducing agents: H2 / Ni 140oC Na / ethanol BACK
From Haloalkane Further substitution give mixture of products. BACK
Physical Properties • With “fishy”smell. Soluble in water due to H-bond formation. Aromatic amine are liquid with high boiling point with a characteristic fishy smell and insoluble in water.
Chemical Properties Basic character Basic strength depends on the availability of lone pair e- on N and solubility in water (stability of the conjugate cation).
Basic character (cont’d) • Inductive effect: • Due to the alkyl group (e-donating group) • 3o > 2o > 1o > NH3 BUT Ease of solvation of protonated amine: Due to the formation of H-bond between N-H and H2ONH3 > 1o > 2o > 3o Overall effect:2o 1o > 3o >NH3
Basic character (cont’d) Comparing ammonia, CH3NH2 & (CH3)2NH, basic strength increased as more alkyl groups attached to the N atom. Alkyl group is e-donating group which makes the lone pair of e- of N more available to co-ordinate with a proton.
Basic character (cont’d) Aromatic amines are less basic strength than aliphatic amines. This is because the lone pair e- of N can overlap with the pi-molecular orbital of the benzene ring. It is thus less available to co-ordinate with a proton.
Resonance Structure of Aniline Electron releasing groups:–OCH3, –CH3increase basic strength Electron withdrawing groups : NO2, –SO3, –COOH, –X decrease it.
Basic character (cont’d) • For acid amide, it is even less basic strength than aromatic amine. This is because the negative inductive effect of the C=O which makes the lone pair e- on N less available. Moreover, lone pair e- on N can ovrelap with p-orbitals of C.
Carbylamine reaction • Aliphatic and aromatic primary amines on heating with chloroform and ethanolic potassium hydroxide form isocyanides or carbylamines which are foul smelling substances.
Test for amine RNH2 + HCl RNH3+Cl- (white fume)
Alkylation (nucleophilic substitution of RX) Such method will give mixture of products.
By Catalytic Hydrogenation The catalytic hydrogenation of nitro group takes place smoothly in presence of finely divided nickel or platinum catalyst
Classification of Amines Characteristic group of primary Amine Primary Amines
Characteristic group of Secondary Amine Secondary Amines
DIAZONIUM SALTS The diazonium salts have the general formula–RN2+X- where R standsforan aryl group and–X ion may be Cl– Br,– HSO4−, BF4− , etc. The stability of arenediazonium ion is explained on the basis of resonance.
Physical Properties of DiazoniunSalts • Benzenediazonium chloride is a colourless crystalline solid. • It is readily soluble in water and is stable in cold but reacts with water when warmed. • It decomposes easily in the dry state. Benzenediazoniumfluoroborateis water insoluble and stable at room temperature.
Importance of Diazonium Salts in Synthesis of Aromatic Compounds • It is clear that the diazonium salts are very good intermediates for the introduction of –F, –Cl, –Br, –I, –CN, –OH,–NO2 groups into the aromatic ring. • Aryl fluorides and iodides cannot be prepared by direct halogenation. • The cyano group cannot be introduced by nucleophilic substitution of chlorine in chlorobenzene but cyanobenzene can be easily obtained from diazonium salt.