Amines
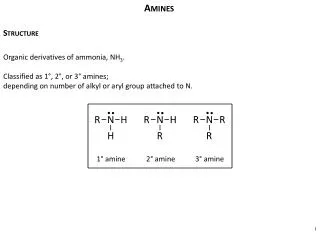
Amines. Structure. Organic derivatives of ammonia, NH 3 . Classified as 1°, 2°, or 3° amines; depending on number of alkyl or aryl group attached to N. 1 ° amine. 2 ° amine. 3 ° amine.

Amines
E N D
Presentation Transcript
Amines Structure Organic derivatives of ammonia, NH3. Classified as 1°, 2°, or 3° amines; depending on number of alkyl or aryl group attached to N. 1° amine 2° amine 3° amine
The amine nitrogen atom is sp3 hybridized and trigonal pyramidal, with bond angles of approximately 109.5°. The amine nitrogen atom has a lone pair, making it both a base and a nucleophile.
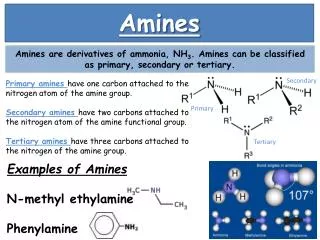
Nomenclature A. Primary Amines They are named as alkylamines (common) or as alkanamines (IUPAC). The complexity of the alkyl groups typically determines which method is used.
B. Secondary and Tertiary Amines They are named as alkylamines (common) with the alkyl groups listed in alphabetical order. The prefixes "di" and "tri" are used if the same alkyl group appears more than once. They are names as alkanamines (IUPAC) with the most complex alkyl group treated as the parent and the simpler alkyl groups treated as substituents, using the locant "N" to identify that they are connected to the nitrogen atom.
C. Ammonium Salts They are derivative of ammonium ion, NH4+.
Physical Properties Amines have polar C—N and N—H bonds and therefore exhibit dipole-dipole interactions. Primary and 2° amines are capable of intermolecular hydrogen bonding because they contain N—H bonds. Since nitrogen is less electronegative than oxygen, these hydrogen bonds are weaker than those between O and H. Amines are more polar and have higher boiling points than alkanes; but are less polar and have lower boiling points than alcohols.
Physical Properties Boiling points of isomeric amines decrease in going from primary to secondary to tertiary amines. Solubility in water can be considerable since H-bonding is possible. Low molecular weight amines (up to ca. five carbons) are quite soluble in water. Amines with low molecular weights, such as trimethylamine, typically have a fishlike odor.
BASICITY OF AMINES Amines are poor acids and require alkyl lithium reagents or alkali metal treatment to form amide ions. They are moderately good bases, leading to the relatively weakly acidic ammonium salts. Alkylamines are slightly stronger bases than ammonia because of the electron-donating inductive effect of the R groups.
Alkylamines differ very little in basicity. Arylamines are much weaker bases than ammonia because the electron pair on N is delocalized.
Alkyl groups on the ring increase basicity, but only slightly. Electron withdrawing groups, especially ortho and/or para to amine group, decrease basicity and can have a large effect.
Amides are much less basic than amines because the electron pair on N is delocalized.
PREPARATION OF AMINES A. Reduction of Nitriles and of Aryl Nitro Compounds
B. Direct Alkylation of NH3 or Amination of RX Sometimes useful to prepare 1° amines Done under restrictive conditions; unwanted multiple alkylation products often obtained
C. Leuckart Reaction Reductive amination of ketone (aldehyde) leading to 1°, 2°, or 3° amines. Imine is temporarily produced but rapidly reduced to the amine under conditions of the reaction
Sodium (or lithium) cyanoborohydride (NaBH3CN) may be used instead of hydrogen as the reducing agent. It reduces C=N but not C=O.
D. The Gabriel Synthesis Clever way to make primary amines from alkyl halides – nucleophilic substitution followed by hydrolysis. The nitrogen-containing nucleophile is potassium phthalimide. As usual, in nucleophilic substitutions, methyl and primary halides give best yields
Acid-catalyzed or base-catalyzed hydrolysis is then performed to release the amine.
E. The Hofmann Rearrangement Another route to primary amines. Solution of bromine in aqueous base is added to 1° amide to form an unstable intermediate which decomposes to 1° amine (minus one carbon).
REACTIONS OF AMINES Aryl Diazonium Salts: Sandmeyer Reactions Primary arylamines react with acidified solutions of sodium nitrite (NaNO2) at 0°C to form aryl diazonium ions Heating the aryl diazonium ions in the presence of various reagents allows the syntheses of various aromatic compounds
The N2 group can be replaced by a nucleophile Sequence = (1) nitration, (2) reduction, (3) diazotization, and (4) nucleophilic substitution
Give the major organic product(s) of each of the following reactions or sequences of reactions
SPECTROSCOPY A. Mass spectrometry Amines with an odd number of N atoms give an odd molecular ion. Alkylamines cleave at the C–C bond nearest the nitrogen to yield an alkyl radical and a nitrogen-containing cation.
B. Infrared Spectroscopy N–H stretching absorptions 3300 to 3500 cm–1. 1° amines show two N—H absorptions whereas 2° amines show one N—H absorption. Amine absorption bands are sharper and less intense than hydroxyl bands.
C. UV spectroscopy Need conjugation.
D. 1H NMR N–H hydrogens appear as broad signals without clear-cut coupling to neighboring C–H hydrogens. In D2O exchange of N–H for N–D occurs, and the N–H signal disappears. Hydrogens on C next to N absorb at lower field than alkane hydrogens.
E. 13C NMR Carbons next to amine N are slightly deshielded, about 30–50 ppm downfield from where they would absorb in an alkane.