Antibody structure and function
Antibody structure and function: http://www.creative-diagnostics.com/Antibody-Structure-and-Function.htm<br><br>

Antibody structure and function
E N D
Presentation Transcript
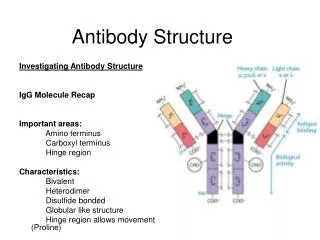
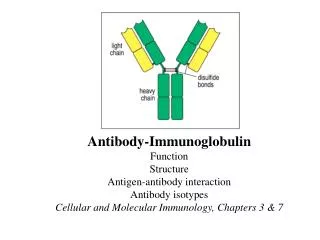
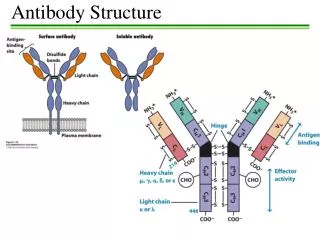
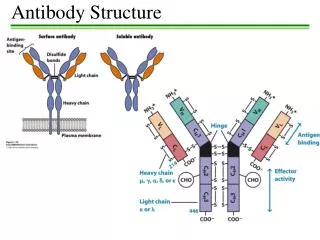
Antibody Structure and Function A typical antibody molecule (IgG, centre) has 12 domains, arranged in two heavy and two light (H and L) chains, linked through cysteine residues by disulphide bonds so that the domains lie together in pairs, the whole molecule having the shape of a flexible Y. In each chain the N-terminal domain is the most variable, the rest being relatively constant. Within the variable (V) regions, the maximum variation in amino acid sequence is seen in the six hypervariable regions (three per chain) which come together to form the antigen-binding site (bottom left in figure). The constant (C) regions vary mainly in those portions that interact with complement or various cell-surface receptors; the righthand part of the figure 1 shows the different features of the C region in the five classes of antibody: M, G, A, E and D. The result is a huge variety of molecules able to bring any antigen into contact with any one of several effective disposal mechanisms. The basic structure (MW about 160 000) can form dimers (IgA, MW 400 000) or pentamers (IgM, MW 900 000). There are species differences, especially in the heavy chain subclasses, which have evolved comparatively recently; the examples shown here illustrate human antibodies. Interestingly, camels and llamas also have antibodies with only heavy chains. These antibodies may be able to attach to some targets not accessible to conventional antibodies, and examples are being tested as possible new ways of preventing infection by viruses such as HIV. Find more about antibody structure and function at Creative Diagnostics. http://www.creative-diagnostics.com/Antibody-Structure-and-Function.htm