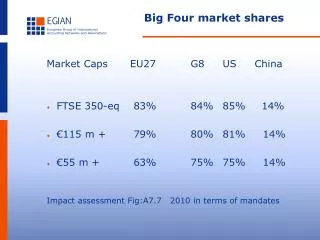
The Big Four
The Big Four. 1. The important Characteristics of Carbon . Forms 4 covalent bonds Forms double and triple bonds Forms long chains and rings Can bind with many other elements Even electron distribution ( nonpolar molecules).

The Big Four
E N D
Presentation Transcript
1. The important Characteristics of Carbon • Forms 4 covalent bonds • Forms double and triple bonds • Forms long chains and rings • Can bind with many other elements • Even electron distribution (nonpolar molecules)
Isomers – molecules that have the same formula but different structures.
2. Macromolecules, Monomers and Polymers(Hint: think of the meaning of the prefixes)
What do these words mean? Micro MACRO
"Poly" Polygons Polyester Polygamy Means...
2. Macromolecules, Monomers and Polymers • Polymer – Smaller organic molecules join into long chains. • Monomer – the individual unit that builds up polymers • Macromolecules – Very large molecules
3. Dehydration synthesis and Hydrolysis • These two terms refer to the processes that forms monomers and polymers. • Dehydration synthesis – A reaction that removes molecules of water to form polymers from monomers (http://nhscience.lonestar.edu/biol/dehydrat/dehydrat.html or http://www.youtube.com/watch?v=UyDnnD3fMaU ) • Hydrolysis – The reaction that adds water to polymers to separate them to their individual monomers.
Three out of the 4 types of biochemical macromolecules can be found on food nutrition labels…
Look at the label to the left. 3 of the 4 macromolecules can be found in foods. FAT • 1____________________ • 2____________________ • 3____________________ (0 grams in this product) Carbohydrates (13 grams in this product) Protein (9 grams in this product)
What is the fourth type of biochemical macromolecule?
4. What are the big four? • Fats (we call them lipids) • Carbohydrates • Proteins • Nucleic acids (DNA and RNA)
When studying these biochemical molecules, we are interested in finding out….. • what they do for living things. • what they generally look like. • what their monomers are. • and how they may help the body gain energy to sustain life. SO, LETS GET STARTED!
5. Carbohydrates • Molecules that form from atoms in 1C:2H:1O ratio • Monomers: Monosaccharides(simple sugars) • Monosaccharides are usually sweet, white powdery substances (such as fructose, glucose) that form rings of carbon atoms.
Monosaccharides in general serve as direct, quick sources of energy for living organisms during cellular respiration, they are building blocks of many polymers • Important monosaccharides: • Glucose • Fructose
Disaccharides – two monosaccharide molecules combine by dehydration synthesis to form disaccharides
Important disaccharides: • Lactose – found in milk sugar • Succrose – table sugar
Polysaccharides – many (tens to hundreds) units of monosaccharides combine by dehydration synthesis • Polysaccharides also separate to monosaccharides by hydrolysis while taking in water.
Important polysaccharides: • Starch – made up of many glucose units, it is an important storage polysaccharide that is found in plant roots and other tissues. It stores monosaccharides that can be broken down later to release useful energy during cellular respiration – ONLY IN PLANTS • Glycogen – also made up of many glucose units, it is an important storage polysaccharide in the liver and animal muscles. It can also be broken down to monomers to release energy during cellular respiration. ONLY IN ANIMALS • Cellulose – also made up of many glucose units. However, in this case the molecule is not easily broken down to its monomers. It is important for providing a rigid structure in plant cell walls.
Chitin – made up of some nitrogen containing monosaccharides. It is an important polysaccharide that provide the solid structure of arthropods and fungi. • http://biomodel.uah.es/en/model3/index.htm
6. Lipids • a diverse group of molecules that are nonpolar and generally do not dissolve in water • They mostly contain carbon, hydrogen, very few oxygen atoms, but some also have phosphorous. • There are three distinct groups of lipids: • Simple lipids • Phospholipids • Sterols
6A. Simple Lipids • Very large molecules that form from 2 different kinds of monomers by dehydration synthesis: • 3 Fatty acids – are long chains of carbon with oxygen at the end (can be saturated and unsaturated) • 1 Glycerol – smaller 3-carbon compound.
Simple lipids are important as storage materials in all living things. They can store twice as many calories as polysaccharides can. Oils (mostly from plants) contain more unsaturated fatty acids, while fats (animals) contain more saturated fatty acids. • Simple lipids also dissolve vitamins • http://biomodel.uah.es/en/model3/index.htm
6B. Phospholipids • Phospholipids – phosphate containing lipids. • Their monomers: 1 glycerol + 2 fatty acids (saturated or unsaturated) + phosphate. These monomers combine by dehydration synthesis • Phospholipids have both polar and nonpolar sections. As a result, they are able to dissolve in both type of solvents as well. • They are important for living things because they form the borders of all cells (cell membranes) and also participate in forming many cell organelles.
6C. STEROLS • Sterols are a highly nonpolar (hydrophobic) group of molecules. They are formed from four rings of carbon atoms. One ring is made up of five carbons, the other three rings are made up of 6 carbons.
What are steroids used for? But some steroids are necessary in certain concentrations.
Cholesterol – important component of the cell membrane, but can be harmful if overdosed in diet. Too much cholesterol can lead to atherosclerosis and other circulatory problems. • Sex hormones – responsible for normal development, sexual function, muscle and body hair formation • Vitamin D – important vitamin for normal bone formation, immune functions • Help to digest fats. • http://biomodel.uah.es/en/model3/index.htm
Monomers: Amino acids – they are small molecules but complex themselves. • Components of amino acids: • Carboxyl group – COOH • Amino group – NH2 • Side chain (labelled R)
The R group can be 20 different kinds of side chains. Because of these side chains we have 20 different types of amino acids in our body. • The R group determines the characteristics of the amino acid • Some amino acids are essential (our body cannot make these, so we need to take these in with our food) while others are nonessential (we can make these)
7B. Combining Amino Acids • Two amino acids can combine at their amino and carboxyl ends by dehydration synthesis. They form a peptide bond together. • The product of this reaction is a dipeptide (2 amino acids), polypeptide (many amino acids) • http://www.johnkyrk.com/aminoacid.html
7C. Protein Structure • Once the proper number of amino acids combine by peptide bond, we get a polypeptide chain – this is the primary structure of a protein (the number and types of amino acids combined to form a protein)
Secondary structure – The long polypeptide chain folds up by using H-bonds. • Alpha helix – If the H-bonds form within the polypeptide chain, a spiral staircase shape is formed. • Beta pleaded sheet – If the H-bonds form between longer stretches of polypeptide chains a long fan shape is formed.
Tertiary structure – all kinds of chemical bonds (covalent, ionic, disulfide bridge, H-bond, van der Waals attraction, dipole-dipole interactions) are used to form the 3 dimensional structure of the polypeptide chain. • If the protein is made up of only one polypeptide chain, than this is the final structural level of the protein
Quaternary structure – If a protein is made up of more than one polypeptide chains, they are connected by various bonds during the quaternary structure of the protein. • http://www.johnkyrk.com/aminoacid.html • http://www.youtube.com/watch?v=lijQ3a8yUYQ
7D. Protein Function • Enzymes – catalyze reactions in the body • Structural proteins – support various structures (cocoon, hair, horns feathers, spider silk) • Storage proteins – proteins in milk, eggs, seeds • Transport proteins – like hemoglobin, carry molecules • Hormones – regulate growth, development, metabolism • Receptors – detect signals from the environment • Motor proteins – like muscle, move organisms or substances inside • Defensive proteins – antibodies that fight against bacteria and viruses • http://www.youtube.com/watch?v=Mszlckmc4Hw&feature=related Prions: http://learn.genetics.utah.edu/content/begin/dna/prions/
7E. Denaturation of proteins • Proteins are very sensitive to a wide range of environmental factors. • Denaturation: When the protein structure is destroyed because of an environmental factor. • Proteins are denatured if: • pH – too high or too low pH • Temperature is too high • Concentration of certain salts is too high
8A. Nucleic Acids – Their Monomers • Macromolecules with complex monomers called nucleotides • Nucleotides are formed from three smaller components: • Monosaccharide with 5 carbon atoms – ribose or deoxyribose • Phosphate ion (PO43- • N-containing bases (Adenine (A), guanine (G), cytosine (C) and thymine (T)
The function of nucleotides is to build up nucleic acids, but they can also act as energy and hydrogen carrying molecules in different biological processes.
8B. DNA (Deoxyribonucleic Acid) • One polymer of nucleotides is DNA that is formed by dehydration synthesis. • DNA has two polynucleotide chains combined with H-bonds at the base section of the nucleotide. The nucleotides that form the H-bond follow the base pairing rule: Adenine binds with thymine (A = T) and guanine binds with cytosine (G = C) • The rest of the nucleotides form the sugar-phosphate backbone • The sugar is always deoxyribose in DNA
DNA is used by all living cells as a storage molecule of their genetic information. DNA is able to copy itself and pass on the genetic material when a cell reproduces. • http://www.pbs.org/wgbh/nova/evolution/darwin-never-knew.html (at 34:00 minutes)
8C. Ribonucleic Acid • One polynucleotide chain, formed by dehydration synthesis from nucleotides. • The bases include adenine, guanine, cytosine and uracil • The sugar is ribose • RNA forms only a single chain of many nucleotides • Its function is to carry small segments of genetic information in the cell