Hydrogen from Algae Nanotechnology Solutions
Hydrogen from Algae Nanotechnology Solutions . Foothill College Bio-Nano-Info Program. Energy from the Early Earth. Energy Metabolism. Hydrogen Metabolism. H 2 S 2H + S H 2 O H + OH H 2 2 H + 2e - In photosynthesis (simplified): H 2 0 H + OH + 2e -


Hydrogen from Algae Nanotechnology Solutions
E N D
Presentation Transcript
Hydrogen from Algae Nanotechnology Solutions Foothill College Bio-Nano-Info Program
Hydrogen Metabolism • H2S 2H + S • H2O H + OH • H2 2 H + 2e- In photosynthesis (simplified): • H20 H + OH + 2e- • 2H + CO2 CH2O • OH + OH H2O + O • 2O + 2e- O2
Timeline for development of the major life forms.From a course site by Robert Huskey, U. Virginia Life on Earth
Hydrogenase • Biological cleavage of H2 is a common metabolic process in prokaryotes and lower eukaryotes and is catalyzed by two major classes of enzymes the [NiFe]- and the [Fe]-hydrogenases. • Three distinct [NiFe]-hydrogenases of Ralstonia eutropha (formerly Alcaligenes eutrophus) are in the center of this project, the regulatory (RH), the NAD-linked (SH) and the membrane-bound (MBH) hydrogenase
Fossilized Blue Green Algae These filaments are believed to be the fossilized imprints of blue-green algae, one of the earliest life forms. They occur in the Bitter Springs Formation in Australia and are about 850 million years old.
Green Algae at Work Making H2 Algal cell suspension / cells Thylakoid membrane
In Vitro Photo-Production of H2 Yellow arrow marks insertion of hydrogenase promoter. Right side exp. optimized for continuous H2 production.
Production of H2 From Algae http://www.eere.energy.gov/hydrogenandfuelcells/pdfs/iic2_lee.pdf
H2 Energy Calculations Assumptions were made that 10 micro mole of H2 can be produced per hour (roughly 50% of peak maximum but extended for an hour) per mg of chlorophyll. Additionally, a density of 10% of the top 1 cm (or 100% of top mm) of the system would be populated by chlorophyll, for a density of 1 mg chlorophyll per square cm of collector. This leads to 10,000 cm multiplied by 10 mg chlorophyll per centimeter for a total of 100,000 mg chlorophyll. Multiplying 100,000 mg chlorophyll by 10 micromole H2 generated per hour per mg chlorophyll yield 1 mole of hydrogen gas per square meter per hour. Combusting one mole of H2 with one half mole of oxygen (H2 + ½ O2 H2O) yields 286 KJoules or 68 Kcal. Using any of the following conversions yields KWatt hours or watts from this reaction: 1 calorie = 4.184 Joules 1 calorie = 0.0011622 KwHr 1 Joule = 0.0002778 Watt hours 1 K Joule = 0.2778 watts 286 KJoules X 0.2778 Watts / KJoules = 79 Watts 68,355 calories X 0.0011622 KwHr per calories = 79 KwHr On first pass, it appears that 1 square meter of hydrogen producing algae (modified for continuous hydrogen production) yields about 79 watts, or enough to run a 75 watt light bulb at full power.
ORNL Project Road Map • Year 1- Design and construction of DNA sequence coding for polypeptide proton channel • Year 2 - Genetic transfer of hydrogenase promoter-linked polypeptide proton-channel DNA into algal strain DS521 • Year 3 - Characterization and optimization of the polypeptide proton-channel gene expression • Year 4 - Demonstration of efficient and robust production of H2 in designer alga (ready for next phase - scale-up and commercialization)
Genetic / Biochemical Engineered H2 Bacterium • Sequence coding for polypeptide proton channel – create gene for proton pump • Genetic transfer of hydrogenase promoter-linked polypeptide proton-channel DNA into algal genome – express pump with H2 • Characterization and optimization of the polypeptide proton-channel gene expression
Proposed Engineered H2 Bacterium http://gcep.stanford.edu/pdfs/tr_hydrogen_prod_utilization.pdf
Polypeptide Proton Channel • Protons that build up from cleavage of H2O into H atoms repress hydrogenase reaction • Need to pump hydrogen atoms away from the photosynthetic reaction core, and into storage • Hydrogen storage in a carbon nanotube can be the first stage in a nano-structure fuel cell • Platinum doped carbon nanotubes might be an integrated device: storage, fuel cell, and battery
Membrane Bound Protein Pumps Proton and ion pumps consume a lot of cellular energy Nano-channels could be useful
Nanotubes / Nanohorns The electrical properties of nanotubes / nanohorns can change, depending on their molecular structure. The "armchair" type has the characteristics of a metal; the "zigzag" type has properties that change depending on the tube diameter—a third have the characteristics of a metal and the rest those of a semiconductor; the "spiral" type has the characteristics of a semiconductor.
Nanotube Properties http://nanotech-now.com/nanotube-buckyball-sites.htm
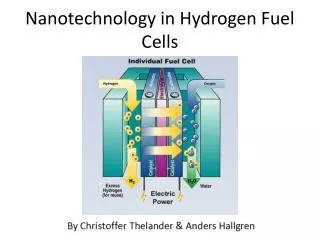
Hydrogen Fuel Cell Basics http://micro.magnet.fsu.edu/primer/java/fuelcell/
Hydrogen Fuel Cell Diagrams Schematic representation of a composite electrode for low temperature fuel cells Schematic representation of themembrane electrode assembly http://www1.physik.tu-muenchen.de/lehrstuehle/E19/research/pefc.html
Summary • Hydrogen metabolism is ancient, and highly conserved in hydrogenase / photosynthesis • With genetic / biochemical engineering, algae can make H2 in significant amounts • Capturing and wicking of H2 into a carbon nanotube fuel cell / battery is very feasible • A 1 sq. meter collector could power a 500 watt household with ~ 10X technology gain