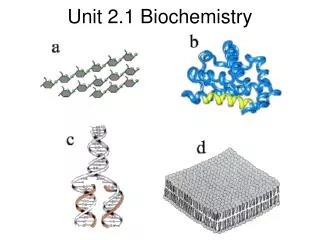
Biochemistry Unit
Biochemistry Unit. PowerPoint #1 – the basics and water…. Matter. Defined as anything that takes up space and has mass Structure of matter refers to its composition – what it is made of – AND how matter is organized…

Biochemistry Unit
E N D
Presentation Transcript
Biochemistry Unit PowerPoint #1 – the basics and water…
Matter • Defined as anything that takes up space and has mass • Structure of matter refers to its composition – what it is made of – AND how matter is organized… • Properties of matter describe the characteristics and behavior of matter, including changes that matter undergoes • (ex. Phase changes – freezing, melting, sublimation, etc.)
ATOMS • Atoms are the basic building blocks of matter that make up everyday objects. • A desk, the air, even YOUR BODY…all are made up of atoms!
Sections of an Atom • Nucleus – protons and neutrons are found here • Orbitals – electrons are found here
Parts of the Atom – subatomic particles • Electron = negative particle circling the nucleus in orbitals • Proton = positive particle found in the nucleus (# of these defines the atom’s location on the Periodic Table and how it reacts – chemical behavior) • Neutron = neutral particle found in the nucleus
Elements 90 occur naturally; the rest are synthetic or radioactive Are put into “families” on the Periodic Table
We’re more than CHNOPS Look at Table 6.1
Elements: ~25 elements are essential to life What are trace elements? Found in the body in very small amounts, yet play vital roles!
Elements All atoms are the same in an element Represented by chemical SYMBOLS Compounds Combination of different amounts of different elements Represented by chemical FORMULAS Elements & Compounds
Compounds • COMPOUNDS: • A substance made of more than one element • Example: H2O = ? • NOTE: the subscripts tell how many atoms of each element you have -- coefficents are used to indicate the amount of each compound present
QUESTION: • How many ELEMENTS are represented by the water formula? • 2 • How many ATOMS are represented by the water formula? • 3
How many atoms?Try These… • KF • Potassium = __; Fluorine = __ • NaBr • Sodium = __; Bromine = __ • H3PO4 • ?????? = __; ?????? = __; ?????? = __ • CuSO4 • ?????? = __; ?????? = __; ?????? = __ • Pb(NO3)2 • ?????? = __; ?????? = __; ?????? = __
KF • Potassium = 1; Fluorine = 1 • NaBr • Sodium = 1; Bromine = 1 • H3PO4 • Hydrogen = 3 • Phosphorus = 1 • Oxygen= 4 • CuSO4 • Copper = 1 • Sulfur = 1 • Oxygen = 4 • Pb(NO3)2 • Lead = 1 • Nitrogen = 2 • Oxygen = 6
Chemical Equations – represent what is taking place when substances react… 2H2 + O2 2H2O • Reactants: substances that undergo chemical reactions. • Products: substance formed by chemical reactions. • Coefficient: shows the # of molecules of each substance • Subscript: the # of atoms in each element in the molecule • Ex. 2H2O = two molecules of water # of atoms: H= 4 O=2 ( 6 total) • Ex #2: C12H22O11 = One molecule of sucrose, # of atoms: 45 • Ex: #3 4C12H22O11 = 4 molecules of sucrose, # of atoms: 180
Equations con’t • Equations must always be balanced!! Again, matter is neither created nor destroyed…….it is only rearranged. • Therefore, the same numbers of atoms on the reactant side MUST equal the number of atoms on the product side.
Balance each equation if necessary • a. N2(g) + H2(g) NH3 (g) • b. CaCO3 (s) CaO (s) + CO2 (g) • c. Se (s) + O2(g) SeO3 (g)
Answers a. N2(g) + 3 H2(g) 2 NH3 (g) b. CaCO3 (s) CaO (s) + CO2 (g) c. 2 Se (s) + 3 O2(g) 2 SeO3 (g)
Bond – to join atoms together The Octet Rule: • atoms form bonds to achieve a Noble Gas electron configuration (want a full outer orbital for stability) • Atoms that achieve this configuration have eight electrons in the outermost orbital – the VALENCE SHELL • Exceptions to this are hydrogen and helium – these are looking for 2 in outer orbital.. Chemical Bond: Force that holds 2 atoms together; each atom is looking to fill its outer orbital for stability
Electrons in the orbitals… • The formula for determining the number of electrons in an orbital is 2n2 where n is the shell number • First orbital can only hold 2 e- Octet rule applies for additional orbitals… • Second orbital can hold 8 e- • Each increasing orbital can hold MORE electrons (s,p,d,f, etc) , but the outermost VALENCE SHELL can never hold more than 8….
Types of bonds • Ionic bonding – bonding that involves a transfer of electrons (one atom gains electrons and one atom loses electrons) – called ions after lose or gain! • Positive charge – cations • Negative charge -- anions compounds are formed • Ex. Na+ + Cl- NaCl
Types of Bonds • Covalent bonding - occurs when atoms share electrons; these atoms have high electron affinity (they attract electrons strongly) and high ionization energy • Molecules are formed • Ex. Water, methane
Hydrogen “Bonds” – really just attractions! • These occur between molecules (or even in different parts of the same molecule). • They are NOT true bonds; rather, are weak attractions between an electronegative atom and a hydrogen already taking part in a second polar covalent bond. • Individually, hydrogen bonds break easily; collectively, they work to stabilize structure. Ex. DNA molecule, water
Bond ranking in terms of individual strength: • Covalent – strongest, “marriage” • Ionic – medium strength, “engagement” • Hydrogen “bond” – weakest, “blind date” *BUT, hydrogen bonds as a GROUP are very supportive and provide structure…
Solutions and Suspensions Solutions and Suspensions are types of mixtures: Solutions are mixtures of two or more substances in which the molecules of the substances are evenly distributed.. Suspensions: mixtures of liquid and nondissolved materials, so there is unequal distribution of materials here…
Solutes and Solvents • To make solutions and suspensions, there are 2 components: Solutes and Solvents • Solutes are the SOLID PARTICLES that are added – *if they dissolve, it is a solution *if they don’t dissolve, it is a suspension • Solvents are the liquids that provide the support or do the dissolving • Water is very common solvent…but it is INCREDIBLY UNIQUE!!!
Water Properties • Powerful, versatile solvent – due to polarity • Cohesive behavior • High Surface Tension • High Specific Heat • High Boiling Point – High Heat of Vaporization • Good evaporative coolant • High freezing point and lower density as a solid than a liquid (density anomaly)
Solvent Capabilities: • Water makes a GREAT solvent because it is a polar molecule – demonstrates unequal sharing of electrons in the covalent bonds The Oxygen is much bigger than the Hydrogens, so the electrons spend more time near the Oxygen, making that end of the molecule appear more negative and the Hydrogens ends appear more positive…
Polar water molecules can Surround and Separate… Forms a hydration shell – ions are surrounded by the water molecules and float away into solution (dissolving)
Hydrophobic and hydrophilic • Hydrophilic substances LIKE water and have a high affinity for it (are attracted to it). Ex. Cotton fibers used in bath towels Hydrophobic substances REPEL water and are NOT attracted to it – won’t mix! Ex: oil in water
Cohesion • Defined as the attraction of water to other water molecules. • Results from hydrogen bonding – temporary associations between molecules with atoms already bonded with other atoms, but “attracted” due to the polarity… those dang “blind dates ” again!
Cohesion and Adhesion • Adhesion is the attraction of different substances to one another (Ex. Water to side of straw) • Cohesion and adhesion work together to give capillarity or capillary action – the ability of water to spread through fine pores or to move upward through narrow tubes against the force of gravity.
Surface Tension • Measure of how difficult it is to stretch or break the surface of a liquid. • Orgs use to stand, walk, or run on water without breaking the surface – Ex. water strider • Due to MANY hydrogen bonds working together at the same time
Water is resistant to temperature changes • Two properties that are related to this: • Specific Heat • Heat of Vaporization
Specific Heat • Def: Amount of heat that must be absorbed or lost for 1 gram of that substance to change by 1 degree Celsius. • High specific heat of water is due to hydrogen bonding – H-bonds tend to restrict molecular movement, so when add heat energy to water, must break bonds first rather than increase molecular motion. • That’s why it takes a LONG time for water to boil!
Heat of Vaporization • Def: Quantity of heat a liquid must absorb for 1 gram of it to be converted from the liquid to the gaseous state. • Again, caused by hydrogen bonds… • Once water boils, it takes quite a while for it to cool back down again, and will continue to “steam” for a long time, too!
Evaporative Cooling • As a liquid evaporates, the surface of the remaining liquid cools down. • Contributes to the stability of temperature in lakes and ponds. • Provides mechanism for cooling organisms – sweating
Freezing • Normally, density increases as temperature decreases. (occurs because molecules are moving more slowly and so, spaces between them decrease, leading to more molecules in same volume)
Density Anomaly • At 4 degrees Celsius – water molecules come so close together that every one of them can form H-bonds simultaneously with 4 other molecules. • Due to geometry of water molecule, they must move slightly apart to maintain the max number of H bonds in a stable structure…so • Below 0 degrees Celsius, an open latticework is formed, allowing air in – thus ice floats on water.
Acids and Bases • certain conditions are vital for chemical reactions to take place – some are better at a low pH (acidic), some are better at a high pH (basic) Ex. • DIGESTION occurs better in acidic conditions, thus stomach acid…. pH – is a logarithmic scale – every number change is a ten-fold change!!!
pH • water is neutral at 7.0 • below 7.0 = acidic • above 7.0 = basic
One last thing on Acids and Bases…. • Many chemicals that we depend on are acids or bases • When they react with one another: the acid loses a H+ and the base loses a OH- and the reaction forms a salt and water …this reaction brings the pH back closer to 7 and is called a buffering reaction…
Example: • HCl + NaOHNaCl + H2O By the way, is this equation balanced?