Exergy Analysis
Exergy Analysis. ME 210 Advanced Thermodynamics. Definitions. Exergy (also called Availability or Work Potential): the maximum useful work that can be obtained from a system at a given state in a given environment; in other words, the most work you can get out of a system

Exergy Analysis
E N D
Presentation Transcript
Exergy Analysis ME 210 Advanced Thermodynamics
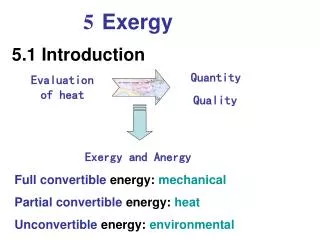
Definitions • Exergy (also called Availability or Work Potential): the maximum useful work that can be obtained from a system at a given state in a given environment; in other words, the most work you can get out of a system • Surroundings: outside the system boundaries • Environment: the area of the surroundings not affected by the process at any point (For example, if you have a hot turbine, the air next to the turbine is warm. The environment is the area of the surroundings far enough away that the temperature isn’t affected.) • Dead State: when a system is in thermodynamic equilibrium with the environment, denoted by a subscript zero; at this point no more work can be done
Example • A coal-fired furnace is used in a power plant. It delivers 5000 kW at 1000 K. The environment is at 300 K. What is the exergy of the added heat? You can use two steps to solve this problem. • Determine the maximum percentage of the heat that can be converted to work. • Using your answer from the first part, determine the maximum work possible. • This is the maximum work output possible between the given state and the dead state, i.e., the heat’s exergy. In this case, 30% of the 5000 kW is unavailable energy—it can’t be converted to work.
Why Study Exergy? • In the last several decades, exergy analysis has begun to be used for system optimization. • By analyzing the exergy destroyed by each component in a process, we can see where we should be focusing our efforts to improve system efficiency. • It can also be used to compare components or systems to help make informed design decisions.
Reversible Work • Wrev (reversible work): the maximum amount of work it’s possible to produce (or minimum necessary to input) in a process between given initial and final states. Note that this is different from an isentropic process where we were given an inlet state and solved for the exit state using s2=s1. Since the exit and inlet states are both fixed, the process is not necessarily isentropic. • What two conditions will cause a process to be isentropic?
Irreversibilities • Irreversibility, I: exergy destroyed; wasted work potential. It represents energy that could have been converted into work but was instead wasted • What are some sources of I? • To have high system efficiency, we want I to be as small as possible.
I, cont. • I=Wrev, out –Wu, out (work output device, like a turbine) OR • I=Wu, in –Wrev, in (work input device, like a pump) • Wu: useful work; the amount of work done that can actually be used for something desirable • Wu=W-Wsurr where W=actual work done
Surroundings Work, Wsurr Here some work is used to push the atmospheric air (the surroundings) out of the way; that work can’t be used for other purposes.
Surroundings Work, Wsurr, cont. • Here Patm helps push the piston in; this is gained work. In a process where the piston goes in and out continually, the surrounding work values cancel out. • What is Wsurr for a control volume?
Second Law Efficiency, hII • Thermal efficiency tells us what we get out compared to what we put in. • The second law efficiency tells us how much we get out compared to the maximum possible we could get out, given the inlet and exit conditions.
Second Law Efficiency, cont. • hth,max=1-TL/TH=1-300/800=0.635 • Say hth=0.45 • hII=0.45/0.625=0.72 • We want a high hth and hII • Another way to look at this: for a work output device hII=Wu/Wrev
Second Law Efficiency, cont. • A general definition:
Three Efficiency Definitions • The second two are defined for work OUTPUT devices
Example • A freezer is maintained at 20°F by removing heat from it at a rate of 75 Btu/min. The power input to the freezer is 0.70 hp, and the surrounding air is 75°F. Determine a) the reversible power, b) the irreversibility, an c) the second-law efficiency of this freezer. Ref: Cengel & Boles, Thermodynamics, An Engineering Approach, 4th edition, Mc-Graw Hill, 2002.):
Exergy • We can calculate the exergy, X (work potential) at a given state. The work potential is a function of the total energy of the system. • (remember that in a control mass, there will be no flow work) • XKE (exergy due to kinetic energy): V2/2 (on a per unit mass basis • XPE: gZ • Xinternalenergy: u-uo+Po(v-vo)-To(s-s0) • To see a derivation of this last equation, see the appendices on the web site. The “o” stands for the dead state (atmospheric conditions). If a piston is at atmospheric pressure and temperature (the dead state), it can’t do any work.
Exergy of a Closed System • Exergy of a closed system, per unit mass j, can be found be adding all the terms • This gives us the maximum work we could possibly get out of a system. • Usually we will be more interested in the change in exergy from the beginning to end of a process. • For a closed system,
For a control volume • Xcv=Xclosed+Xflowwork • y=Xcv/m (exergy per unit mass) • Xflow work=Wflow-Wagainstatmosphere=Pv-Pov • Now combine terms: u+Pv=h; uo+Povo=ho
Change in exergy • If we only have one fluid stream • If we have multiple streams
Exergy Balance • We will use these equations in an exergy balance to solve for such quantities as reversible work or exergy destroyed. • Xin-Xout-Xdestroyed=DXsys • Xdestroyed is potential work that was destroyed due to irreversibilities like friction. • Exergy can be transferred (Xin-Xout) by heat, work, and mass flow
Exergy Transfer by Heat Transfer • As we add heat to a system, we increase its ability to do work. • See Appendix B on web for a discussion of how to deal with cold sinks.
Exergy Transfer by Work and Mass Flow • If we do work on a system, we increase its ability to do work. • Xwork=W-Wsurr for boundary work • Xwork=W for all other kinds of work • Remember • Xmass=my
Xdestroyed • Xdestroyed=I=ToSgen • See Appendix C on the web for a derivation. • Review from ME 297 • DSsys=Sin-Sout+Sgen
Entropy Generated, Sgen • For a steady-state control volume, this leads us to • For a control mass, this becomes • Here Tk is the temperature of the heat source or heat sink (not the system temperature).
Final Equation for DXsys for control mass • Terms in [ ] are W-Wsurr=Wu • If we want to find Wrev, then ToSgen=0 and Wu=Wrev • Note that if heat transfer is to/from the surroundings, the Q term drops out.
Example • A 12-ft3 rigid tank contains R-134a at 30 psia and 40% quality. Heat is transferred now to the refrigerant from a source at 120°F until the pressure rises to 60 psia. Assuming the surroundings to be at 75°F, determine a) the amount of heat transfer between the source and the refrigerant and b) the exergy destroyed during the process. Ref: Cengel and Boles
Final Equation for DXsys for control volume For multiple fluid streams, unsteady flow: For one fluid streams, steady flow: To find Wrev, set Sgen=0. If adiabatic, Q=0.
Set up the following problems. • Refrigerant at T1 and P1 is throttled to a pressure of P2. Find the reversible work and exergy destroyed during this process. The atmosphere has a temperature of To. • Air at T1 and P1 with a velocity of V1 enters a nozzle and exits at P2 and T2 with a velocity of V2. There is a heat loss Q from the nozzle to the surroundings at To. Find the exergy destroyed during this process. • Air enters a compressor at ambient conditions (To and Po) and leaves at P2 and T2. The compressor is deliberately cooled, and there is a rate of heat loss of Q to the surroundings. The power input to the compressor is PWR. Find the rate of irreversibility, I, for this process.
Example • See handout