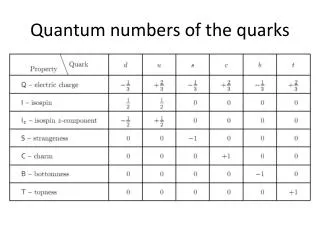
Quantum Numbers
Quantum Numbers. Rules of Electron Location and Orbital Filling Order. If the arrangement of the electron is known, a great deal of information can be predicted about the element. Quantum Numbers At the conclusion of our time together, you should be able to:.

Quantum Numbers
E N D
Presentation Transcript
Quantum Numbers Rules of Electron Location and Orbital Filling Order
If the arrangement of the electron is known, a great deal of information can be predicted about the element
Quantum NumbersAt the conclusion of our time together, you should be able to: • List and define each of the 4 quantum numbers. • Relate these numbers to the state, city, street and home address for the electron. • Give the maximum number of electrons for each level and sublevel. • Draw the basic shape of the 4 sublevels.
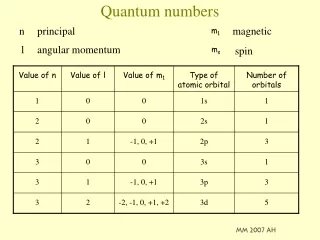
Principal Quantum Number - n Symbol = n Represents the main energy level of the electron and its distance from the nucleus Equation: 2n2 - shows how many electrons can be in each energy level (e.g. 3rd energy level: 2(3)2 = 18 total possible e- in this energy level) Your turn: How many electrons in the 4th energy level? 32
Why God Made MomsAnswers given by 2nd grade school children to the following questions:Why did God give you your mother and not some other mom? 1. We're related. 2. God knew she likes me a lot more than other people's mom like me.
Principal Quantum Number - n • In an address analogy, this would be the state in which the electron would probably be found. Presently, we can find electrons in 7 states. • Values are 1-7 • Ex. = 1s1 (the electron configuration for H) Principal Quantum number = 1
The First of 4 Quantum Numbers for the One Electron of Hydrogen
The Second Quantum Number - l • This number describes sublevels, shapes of these sublevels and is the Angular momentum quantum number. The number of sublevels (shapes) in an energy level equals the value of n, the principle quantum number. The first 4 sublevels are named s, p, d, f. • Each sublevel has a unique shape:
Shape of the “s” Orbital • s for "Sphere": the simplest shape, or shape of the simplest atoms like hydrogen and helium • Electrons don't interfere with, or block, each other from the pull of the nucleus - ball shape • Each energy level has an "s" orbital at the lowest energy within that level
Heisenberg Uncertainty Principle It is impossible to determine both the position and the momentum of an electron at the same time. Why???
Shapes of the “p” Orbitals • p for "Peanut/Petal": a more complex shape that occurs at energy levels 2 and above
Shapes of the “d” Orbitals • d for "Double Peanut/Petal": a complex shape occurring at energy levels 3 and above • The arrangement of these orbitals allows for "s" and "p" orbitals to fit closer to the middle/nucleus
Shapes of the “f” Orbitals • f for "Flower": bizarre-shaped orbitals for electrons of very large atoms • electrons filling these orbitals are weakly attached to the atom because they are so far away from the pull of the nucleus
The Second Quantum Number - l • In the address analogy, this would be the city in which the electron would probably be found. In the first state there would be 1 city, in the second state there would be 2 cities... • This number is from the formula: 0 to (n-1)
The Second Quantum Number - l • The 2nd Quantum Number = 0 to (n-1) • Ex. = 1s1 , s sublevel number is 0 to (1-1) = 0 • p sublevel number is, 0 to (2-1) = 1 • d sublevel number is, 0 to (3-1) = 2 • f sublevel number is, 0 to (4-1) = 3 • Therefore: • For s, l = 0 • For p, l = 1 • For d, l = 2 • For f, l = 3
The Second of 4 Quantum Numbers for the One Electron of Hydrogen Ex. = 1s1, 0 to (1-1) = 0 Second Quantum number for Hydrogen = 0
Why God Made MomsAnswers given by 2nd grade school children to the following questions:If you could change one thing about your mom, what would it be? 1. She has this weird thing about me keeping my room clean. I'd get rid of that. 2. I'd make my mom smarter. Then she would know it was my sister who did it, not me. 3. I would like for her to get rid of those invisible eyes on the back of her head.
The Third Quantum Number - m • This number describes orientations of the orbitals that each sublevel can have and is the Magnetic quantum number. • s has one orientation, • p has three orientations , • d has five orientations and • f has 7 orientations. • We’ll see how we got these orientation numbers in just a moment.
The Third Quantum Number - m In the address analogy, this would be the street on which the electron would probably be found. In the first state there is one city. In this first city there would be one street. In the second state there are two cities. The first city with its one street and a second city with its 3 streets for a total of 4. In the 3rd state would have the previous 4 streets plus 5 more streets from the 3rd city for a total of 9.
The Third Quantum Number - m • 3rd Quantum Number = –l to 0 to +l • We know what the shape looks like from the second Quantum Number, now we know how many orientations of these shapes each sublevel has by plugging the l value into the formula above.
The Third Quantum Number - m Value = –l to 0 to +l, therefore: • s = ___ • 0 Or just 1 orientation
The Third Quantum Number - m Values are –l to 0 to +l, therefore: • p = ___ ___ ___ -1 0 +1 • Or 3 orientations
The Third Quantum Number - m Values are –l to 0 to +l, therefore: • d = ___ ___ ___ ___ ___ • -2 -1 0 1 2 • or 5 orientations
The Third Quantum Number - m Values are –l to 0 to +l, therefore: • f = ___ ___ ___ ___ ___ ___ ___ • -3 -2 -1 0 1 2 3 • or 7 orientations
This is in every kitchen… The Kitchen Zinc-Zn
The Third Quantum Number - m Let’s Review the Values for m: • s = ___ p = ___ ___ ___ • 0 -1 0 +1 • d = ___ ___ ___ ___ ___ • -2 -1 0 1 2 • f = ___ ___ ___ ___ ___ ___ ___ • -3 -2 -1 0 1 2 3 • Ex. = 1s1 = 0 Third Quantum number from s = 0, or 1 shape
The Third of the 4 Quantum Numbers for the One Electron of Hydrogen
The Fourth Quantum Number - s • This number describes the spin of the electrons in an orbital. There can be two electrons in each orbital as long as they are spinning in opposite directions. • In the address analogy, this would be • the house number of the electron. • There can only be 2 houses on each street • Values are +1/2 = clockwise spin • -1/2 = counter clockwise spin
The Fourth Quantum Number - s • Ex. = 1s1 , spin is up or +1/2 The Fourth Quantum number from superscript 1 = +1/2
The Fourth of the 4 Quantum Numbers for the One Electron of Hydrogen
Quantum # Summary for Hydrogen • 1s1 • Hydrogen has one electron spinning in a clockwise direction in the first energy level that has one orbital and only one orientation in its s sublevel shape which is spherical. • The 4 quantum numbers for H are: • 1, 0, 0, +1/2
The teenager fell into a frozen lake and was very… Gold - Au
Quantum NumbersLet’s see if you can: • List and define each of the 4 quantum numbers. • Relate these numbers to the state, city, street and home address for the electron. • Give the maximum number of electrons for each level and sublevel. • Draw the basic shape of the 4 sublevels.
How do we know when an electron has moved from an excited state to the ground state? The electron will • Release a photon. • Release a specific amount of energy. • Release a specific color. • Release a quantum of energy. • All of the above are correct.
What does the second quantum number (l) describe? • Orbital shape. • Energy level. • Electron spin. • Orbital orientation.
Which quantum number has values of +½ or –½? • Orbital shape. • Energy level. • Electron spin. • Orbital orientation.
Which of the following is not a possible orbital shape? • s • p • z • f
What is currently the highest possible principal quantum number an electron can have? • No limit • 5 • 6 • 7
How many electrons will the 4th energy level hold? • No limit • 8 • 18 • 32 • 50
The p sublevel would look like a • sphere • petal • double petal • flower • mess
Quantum NumbersAt the conclusion of our time together, you should be able to: • Continue to relate Quantum Numbers to the state, city, street and home address for the electron. • Give the 4 quantum numbers for every electron of every atom on the periodic table.
If the arrangement of the electron is known, a great deal of information can be predicted about the element
Let’s Try Helium • H is 1s1 • He has 2 electrons, can we add another electron spinning in the other direction in the first energy level of the s sublevel with its 1 spherical orbital? • Yes, He is 1s2 • He has 2 electrons spinning in opposite directions in the s sublevel with its spherical shape and its 1 orientation.
Remember the Quantum # Summary for Hydrogen • 1s1 • Hydrogen has one electron spinning in a clockwise direction in the first energy level that has one spherical orbital in its s sublevel. • The 4 quantum numbers for H are: • 1, 0, 0, +1/2
The 4 Quantum Numbers of Helium • No two electrons in an atom can have the same set of four quantum numbers. • What principle?? • Pauli Exclusion Principle • Therefore, the second electron that He has must have a different set of 4 Quantum Numbers. • What would they be?? • 1, 0, 0, -1/2