Diffusion Evolution of the distribution function Boltzmann equation velocity modulation
440 likes | 605 Views
Diffusion Evolution of the distribution function Boltzmann equation velocity modulation. Transport properties. mobility. Ohm's law. Find the conductivity of a resistor. E. Conductivity of a plasma. Neutral particle. Electron. Diffusion. Diffusion. Recall the RC transmission line

Diffusion Evolution of the distribution function Boltzmann equation velocity modulation
E N D
Presentation Transcript
Diffusion Evolution of the distribution function Boltzmann equation velocity modulation
Transport properties mobility Ohm's law
E Conductivity of a plasma Neutral particle Electron
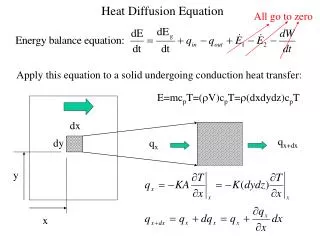
Diffusion Recall the RC transmission line from 55:070.
Diffusion units
delta function source impulse function source
P. D. E. O. D. E.
Response to a delta function source
I S U hot stove - constant source
Response to A constant voltage source
v x Harmonic oscillatorhttp://www.kettering.edu/~drussell/Demos/SHO/mass.html Phase space
Boltzmann equationhttp://grus.berkeley.edu/~jrg/ay202/node32.html Ludwig Eduard Boltzmann (1844-1906). The physicist whose greatest achievement was in the development of statistical mechanics, which explains and predicts how the properties of atoms determine the visible properties of matter such as viscosity, thermal conductivity, and diffusion. He is reputed to have smuggled wine into the Faculty Club during his 1904 visit to Berkeley--at that time Berkeley was a dry town
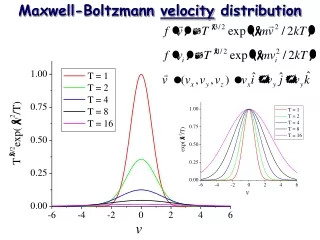
Boltzmann equation How does a distribution function evolve in non-equilibrium systems? At a later time Difference is due to collisions.
v x Boltzmann equation
Water bag distribution Same area
v v v x x x Boltzmann equation t = 0 t > 0 t = shock
Velocity modulationhttp://www2.slac.stanford.edu/vvc/accelerators/klystron.html Pipe organ
Velocity modulationhttp://www.google.com/search?hl=en&client=firefox-a&rls=org.mozilla%3Aen-US%3Aofficial&hs=aBD&q=Applegate+diagram&btnG=Search Applegate diagram
v x Phase space interpretation of velocity modulation t = 0 t = shock
Velocity modulationhttp://www2.slac.stanford.edu/vvc/accelerators/klystron.html Pipe organ klystron
Summary of physics principles • Waves and particles are related – photo electric effect
Summary of physics principles • Schrödinger equation gives us empty states and quantum numbers • Fermi function tells us if the state is filled • Boltzmann equation describes how the distribution of states evolves in space and time • Velocity modulation helped win World War II and demonstrates the Boltzmann equation
Energy bands • With more than one atom, one has to inquire about possible interaction between individual particles. • Think of two race cars (or two witches) – the one behind uses less energy if it is following very closely behind the first one. • Splitting of individual energy levels yielding a band.
Energy bands • Probability density functions from two adjacent atoms in close juxtaposition causes interaction and splitting of the lowest state yielding a band. separated close together band
Energy bands • Valence band – the top most energy band containing electrons • Conduction band – the energy band just above the valence band • Electrons in the conduction band can move from one location to another
Why did the chicken cross the road? • Aristotle: It is the nature of chickens to cross roads. • Issac Newton: Chickens at rest tend to stay at rest, chickens in motion tend to cross roads. • Albert Einstein: Whether the chicken crossed the road or the road moved beneath the chicken depends on your frame of reference. • Werner Heisenberg: We are not sure which side of the road the chicken was on, but it was moving very fast. • Wolfgang Pauli: There already was a chicken on this side of the road.