Lecture 15: The Hydrogen Atom
440 likes | 873 Views
Lecture 15: The Hydrogen Atom . J.J. Thomson’s Plum Pudding Model of the Atom (1897). He proposed that the electrons are embedded in a positively charged ‘pudding’. Rutherford’s a Scattering Experiment (1911).

Lecture 15: The Hydrogen Atom
E N D
Presentation Transcript
J.J. Thomson’s Plum Pudding Model of the Atom (1897) He proposed that the electrons are embedded in a positively charged ‘pudding’
Rutherford’s a Scattering Experiment (1911) He found that, once in a while, the a-particles were scattered backwards by the target video clip
Discovery of the Atomic Nucleus To explain the backscattering, the positive charge must be concentrated in a small region
Rutherford’s Solar System Model of the Atom The atom consists of electrons orbiting around a small but dense central nucleus
Hydrogen Atom is Unstable? • It is known that accelerating charges emit radiation • Thus, electron should emit radiation, lose energy and eventually fall into the nucleus! • Why doesn’t this happen? Shows that something was wrong with this model of the hydrogen atom
Absorption Spectrum of a Gas Dark lines will appear in the light spectrum
Absorption spectrum of Sun Emission spectra of various elements
Balmer’s Formula for Hydrogen • Notice there are four bright lines in the hydrogen emission spectrum • Balmer guessed the following formula for the wavelength of these four lines:where n = 3, 4, 5 and 6
Bohr’s Model of the Hydrogen Atom(1913) He proposed that only certain orbits for the electron are allowed
Bohr’s Empirical Explanation • Electrons can only take discrete energies (energy is related to radius of the orbit) • Electrons can jump between different orbitsdue to the absorption or emission of photons • Dark lines in the absorption spectra are due to photons being absorbed • Bright lines in the emission spectra are due to photons being emitted
Absorption / Emission of Photonsand Conservation of Energy Ef - Ei = hf Ei - Ef = hf
Electron jumping to a higher energy level E = 12.08 eV
Spectrum of Hydrogen Bohr’s formula:
Hydrogen is therefore a fussy absorber / emitter of light It only absorbs or emits photons with precisely the right energies dictated by energy conservation
This explains why some nebulae are red or pink in colour One of the transitions in the Balmer series corresponds to the emission of red light
Schrödinger’s Improvement to Bohr’s Model • Showed how to obtain Bohr’s formula using the Schrödinger equation • Electron is described by a wave function y • Solved for y in the electric potential due to the nucleus of the hydrogen atom
Square Well • Approximate electric (roller coaster) potential by a ‘square well’ • System is then identical to the wave equation for a string that is fixed at both ends
Vibrational Modes of a String fundamental 2nd harmonic 3rd harmonic 4th harmonic
Quantum Numbers • Energy levels can only take discrete values • Labelled by a ‘quantum number’ n, which takes values 1, 2, 3, ... • Each level has energy that increases with n
Ground State (n=1) • Lowest or ground-state energy is non-zero • Electron cannot sit still but must be forever ‘jiggling around’ • Expected from the Heisenberg uncertainty principle
Vibrational Modes of a Rectangular Membrane (1,1) mode (1,2) mode (2,1) mode (2,2) mode Vibrational modes of a circular membrane (drum)
Electron in a Hydrogen Atom • Wave function is like a vibrating string or membrane, but the vibration is in three dimensions • Labelled by three quantum numbers: • n = 1, 2, 3, … • ℓ = 0, 1, …, n-1 • m = -ℓ, -ℓ+1, …, ℓ-1, ℓ • For historical reasons, ℓ = 0, 1, 2, 3 is also known as s, p, d, f
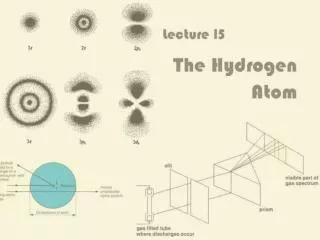
Density of the cloud gives probability of where the electron is located
Another diagram of 2p orbitals Note that there are three different configurations corresponding to m = -1, 0, 1
3d Orbitals Now there are five different configurations corresponding to m = -2, -1, 0, 1, 2
4f Orbitals There are seven different configurations corresponding to m = -3, -2, -1, 0, 1, 2, 3
Summary • Electron does not fly round the nucleus like the Earth around the Sun (Rutherford, Bohr) • Depending on which energy level it is in, the electron can take one of a number of stationary probability cloud configurations (Schrödinger)