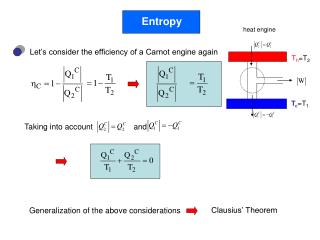
Entropy
Entropy. - In classical thermodynamics the entropy is defined by: - the entropy can be related to S as: we call S the “conventional” entropy. Entropy increase on heat flow.

Entropy
E N D
Presentation Transcript
Entropy - In classical thermodynamics the entropy is defined by: - the entropy can be related to S as: we call S the “conventional” entropy Entropy increase on heat flow The total entropy change of the system when we remove a positive amount of energy U from system 1 and the same amount to system 2: When 1> 2 dS is positive, otherwise negative Law of increase of entropy The total entropy always increases when two systems are brought into thermal contact! Let U10 and U20 be the initial internal energy of system 1 and 2 respectively, and let U=U10+U20 be the total energy. We can write than: Here stands for the energy of system 1 in the most probable configuration when it is in thermal contact with system 2. For the entropy we get: The evolution of the combined system towards the final thermal configuration is realized through intermediate configurations with intermediate energies and entropy. Law of increase of entropy
Laws of Thermodynamics - When thermodynamics is studied as a nonstatistical subject (phenomenology) --> four postulates - These postulates are theoretically based in our statistical formulation. Zeroth law: If two systems are in thermal equilibrium with a third system, they must be in thermal equilibrium with each other. Immediate consequence of the condition for thermal equilibrium First law: Heat is a form of energy and together with work can contribute to the change of the internal energy of the system --> practically the principle of energy conservation. Second law: the law of increase of entropy. If a closed system is in a configuration that is not the equilibrium configuration , the most probable consequence will be that the entropy of the system will increase monotonically in successive instants of time. Many other formulations are known…. Third law: The entropy of a system approaches a constant value (must come in flat with T) as the temperature approaches zero. This constant value might not necessarily be: 0 Entropy as a logarithm:two important consequences of the fact that entropy is defined as the logarithm of the number of accessible states, instead of as the number of accessible states itself: 1. The entropy becomes an extensive parameter 2. The entropy is insensitive to the precision with which the energy of a closed system is defined or fixed. Let D(U) be the number of accessible states per unit energy range. From here: Typically , as for the system of N spins, g(U) is of order 2N, D(U)2N/N, where is the average energy per particle.
Problems: 1. Find why the solution of the Example: “Entropy increase on heat flow” (page 44-45) is not totally correct. In fact it is quite a rough approximation…. 2. We pour a glass of water (m=0.2 kg) at temperature Ti=600 C in a lake with water at temperature T0=100C. Calculate the change in the entropy of the Universe due to this process. The specific heat of water is: cw=4180 J/(Kg.K) 3. Solve problem 5 on page 53. 4. Solve problem 6 on page 54 Extra problem: A famous British duo Flanders and Swan have a nice song about the laws of thermodynamic. You can download it (I suggest to listen it, it is funny) in mp3 format here, and you can get the verses here. Find the scientifically wrong statements in their song, and argue why it is wrong or not complete.