Entropy
Physics 202 Professor Lee Carkner Ed by CJV Lecture -last. Entropy. Entropy. What do irreversible processes have in common? They all progress towards more randomness The degree of randomness of system is called entropy For an irreversible process, entropy always increases

Entropy
E N D
Presentation Transcript
Physics 202 Professor Lee Carkner Ed by CJV Lecture -last Entropy
Entropy • What do irreversible processes have in common? • They all progress towards more randomness • The degree of randomness of system is called entropy • For an irreversible process, entropy always increases • In any thermodynamic process that proceeds from an initial to a final point, the change in entropy depends on the heat and temperature, specifically: DS = Sf –Si = ∫ (dQ/T)
Isothermal Entropy • In practice, the integral may be hard to compute • Need to know Q as a function of T • Let us consider the simplest case where the process is isothermal (T is constant): DS = (1/T) ∫ dQ DS = Q/T • This is also approximately true for situations where temperature changes are very small • Like heating something up by 1 degree
State Function • Entropy is a property of system • Like pressure, temperature and volume • Can relate S to Q and thus to DEint & W and thus to P, T and V DS = nRln(Vf/Vi) + nCVln(Tf/Ti) • Change in entropy depends only on the net system change • Not how the system changes • ln 1 = 0, so if V or T do not change, its term drops out
Entropy Change • Imagine now a simple idealized system consisting of a box of gas in contact with a heat reservoir • Something that does not change temperature (like a lake) • If the system loses heat –Q to the reservoir and the reservoir gains heat +Q from the system isothermally: DSbox = (-Q/Tbox) DSres = (+Q/Tres)
Second Law of Thermodynamics (Entropy) • If we try to do this for real we find that the positive term is always a little larger than the negative term, so: DS>0 • This is also the second law of thermodynamics • Entropy always increases • Why? • Because the more random states are more probable • The 2nd law is based on statistics
Reversible • If you see a film of shards of ceramic forming themselves into a plate you know that the film is running backwards • Why? • The smashing plate is an example of an irreversible process, one that only happens in one direction • Examples: • A drop of ink tints water • Perfume diffuses throughout a room • Heat transfer
Randomness • Classical thermodynamics is deterministic • Adding x joules of heat will produce a temperature increase of y degrees • Every time! • But the real world is probabilistic • Adding x joules of heat will make some molecules move faster but many will still be slow • It is possible that you could add heat to a system and the temperature could go down • If all the molecules collided in just the right way • The universe only seems deterministic because the number of molecules is so large that the chance of an improbable event happening is absurdly low
Statistical Mechanics • Statistical mechanics uses microscopic properties to explain macroscopic properties • We will use statistical mechanics to explore the reason why gas diffuses throughout a container • Consider a box with a right and left half of equal area • The box contains 4 indistinguishable molecules
Molecules in a Box • There are 16 ways that the molecules can be distributed in the box • Each way is a microstate • Since the molecules are indistinguishable there are only 5 configurations • Example: all the microstates with 3 in one side and 1 in the other are one configuration • If all microstates are equally probable than the configuration with equal distribution is the most probable
Configurations and Microstates Configuration I 1 microstate Probability = (1/16) Configuration II 4 microstates Probability = (4/16)
Probability • There are more microstates for the configurations with roughly equal distributions • The equal distribution configurations are thus more probable • Gas diffuses throughout a room because the probability of a configuration where all of the molecules bunch up is low
Multiplicity • The multiplicity of a configuration is the number of microstates it has and is represented by: W = N! /(nL! nR!) • Where N is the total number of molecules and nL and nR are the number in the right or left half n! = n(n-1)(n-2)(n-3) … (1) • Configurations with large W are more probable • For large N (N>100) the probability of the equal distribution configurations is enormous
Entropy and Multiplicity • The more random configurations are most probable • They also have the highest entropy • We can express the entropy with Boltzmann’s entropy equation as: S = k ln W • Where k is the Boltzmann constant (1.38 X 10-23 J/K) • Sometimes it helps to use the Stirling approximation: ln N! = N (ln N) - N
Irreversibility • Irreversible processes move from a low probability state to a high probability one • Because of probability, they will not move back on their own • All real processes are irreversible, so entropy will always increases • Entropy (and much of modern physics) is based on statistics • The universe is stochastic
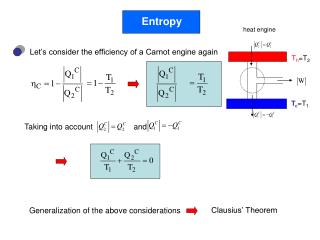
Engines and Refrigerators • An engine consists of a hot reservoir, a cold reservoir, and a device to do work • Heat from the hot reservoir is transformed into work (+ heat to cold reservoir) • A refrigerator also consists of a hot reservoir, a cold reservoir, and a device to do work • By an application of work, heat is moved from the cold to the hot reservoir
Refrigerator as a Thermodynamic System • We provide the work (by plugging the compressor in) and we want heat removed from the cold area, so the coefficient of performance is: K = QL/W • Energy is conserved (first law of thermodynamics), so the heat in (QL) plus the work in (W) must equal the heat out (|QH|): |QH| = QL + W W = |QH| - QL • This is the work needed to move QL out of the cold area
Refrigerators and Entropy • We can rewrite K as: K = QL/(QH-QL) • From the 2nd law (for a reversible, isothermal process): QH/TH = QL/TL • So K becomes: KC = TL/(TH-TL) • This the the coefficient for an ideal or Carnot refrigerator • Refrigerators are most efficient if they are not kept very cold and if the difference in temperature between the room and the refrigerator is small