Dose
Dose. Particles lose energy in matter. Eventually energy loss is due to ionization. An important measure is the amount of energy gained by the material as a particle passes or stops. Energy Gained. Energy transferred describes the kinetic energy gained by charged particles.

Dose
E N D
Presentation Transcript
Particles lose energy in matter. Eventually energy loss is due to ionization. An important measure is the amount of energy gained by the material as a particle passes or stops. Energy Gained
Energy transferred describes the kinetic energy gained by charged particles. Energy imparted is the energy lost by charged particles. Energy Transferred 1.1 2.8 4.2 5.4 10 MeV particle energy 3.6 2.0 0.1 9.0 energy transferred 2.8 3.3 2.8 1.1 energy imparted
Energy transferred Radiant energy into a volume from uncharged particles Radiant energy out tpo uncharged particles (not brem or annihilation) Energy change from mass Energy imparted Start with energy transferred Energy in from charged particles Energy out from charged particles Energy Imparted
Kerma is the energy transferred per unit mass. Kinetic Energy Released per unit MAss Radiative kerma is the energy loss per mass due to brem and annihilation. Collision kerma subtracts the reradiated photons. Net energy transferred per mass Kerma
Absorbed dose or dose is the energy imparted per unit mass. Like kerma dose is based on mean changes in energy. Two units are used. 1 gray (Gy) = 1 J / kg 1 rad = 100 erg / g (older) Absorbed Dose
Dose can be compared to physical effects. Lethality refers to the likelihood that a dose will be fatal. Cell death Whole body death (see graph at right) Lethality Lethality % Dose (cGy) Federation of American Scientists
Measuring the relationship between energy transferred and imparted requires equilibrium conditions. Radiation equilibrium Charged particle equilibrium Looser requirement Equilibrium
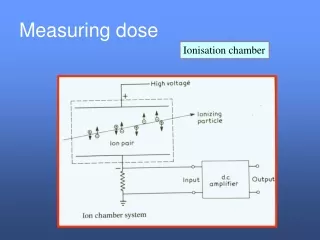
Exposure is defined by the ionization produced by photons. Gammas and X-rays Charge per unit mass in air The original unit is 1 esu / cm3 of dry air at STP (1928). Roentgen (R) R = 2.58 x 10-4 C / kg (1962) Useful Conversion Show that the original roentgen is equivalent to the modern one. Look up constants: Density of air at STP is 0.001293 g / cm-3 1 esu = 3.34 x 10-10 C 3.34 x 10-10 C / 1.293 x 10-6 kg = 2.58 x 10-4 C / kg Exposure
Assume a point source of gammas. Activity C Average photon energy E Consider a sphere or radius r. Fluence through sphere F Mass energy absorption men/r = 2.7 x 10-3 m2/kg Find the dose and exposure rate. 1 R = 0.0088 Gy Rate of energy release: Fluence rate: Dose rate: Exposure rate: Gamma Rate
The effect of radiation on tissue depends on the LET as well as the dose. Higher LET is more damaging. Radiation has a weighting factor based on particle. Factor WR or Q Updated in 1991 In terms of LET LET L (keV / mm in water) < 10; WR = 1 10 – 100; WR = 0.32L – 2.2 > 100; In terms of particle e, g, m; WR = 1 n; WR = 5 – 20 p; WR = 5 a; WR = 20 Radiation Factor
Secondary electrons ejected from atoms are delta rays. Deltas deliver most of the dose Energy less effective if it’s too high Restricted stopping power measures the energy lost up to a limit D. Typical Problem Estimate a cutoff value for irradiating 300 Å viruses. Answer Most energetic delta range should not exceed 300 Å. Find range limit 3 x 10-6 cm Range in water 500 eV is 2 x 10-6 g/cm2 1 keV is 5 x 10-6 g/cm2 Estimate cutoff at 700 eV. Restricted Stopping Power
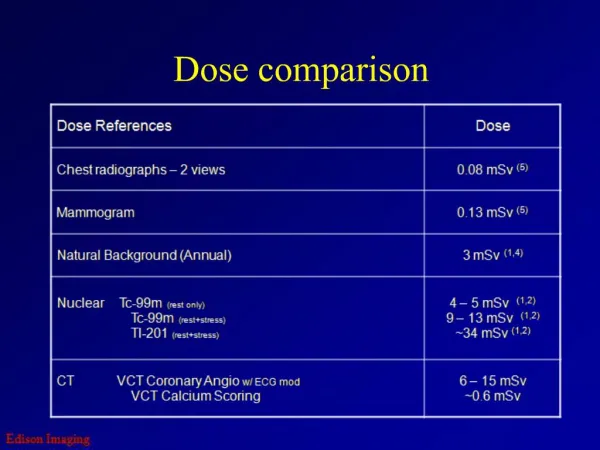
The equivalent dose is a measure that combines the type of radiation and dose. Unit is Sievert (Sv) 1 Gy equivalent Older unit is rem Roentgen equivalent man 1 rad equivalent 100 rem = 1 Sv Natural doses Cosmics: 0.3 mSv / yr Soil: 0.2 mSv / yr Radon: 2 mSv / yr Total natural: 3 mSv / yr Environmental hazards Flying at 12 km: 7 mSv / hr Chest x-ray: 0.1 mSv Mammogram: 1 mSv CT scan: 20 mSv Equivalent Dose
Neutron Interactions • Neutrons present a unique situation for dose determination. • No interaction with atomic electrons • Cross sections vary with target nucleus
Elastic scattering from nuclei is the most important process for neutron energy loss. Assume classical collision Set M = 1 and compare for nuclei Nucleus Qmax/En 1H 1.000 2H 0.889 4He 0.640 9Be 0.360 12C 0.284 16O 0.221 56Fe 0.069 118Sn 0.033 238U 0.017 Neutron Scattering
Neutron weighting factors were variable. WR = 5-20. The factors can be determined from assumed elastic scattering. Example Find WR for 2-MeV neutrons. Average recoil p is 1 MeV Stopping power for 1 MeV p in water is 270 MeV/cm Equal to 27 keV/mm Neutron Dose Equivalent
Inelastic collisions result in a nuclear reaction. Many are endothermic Requires extra energy For example 32S(n,p)32P: Eth = 0.957 MeV 32P 32S + b- Ebmax = 1.71 MeV T = 14.3 days Used to detect exposure Change in rest energy Conservation of energy and momentum Neutron Threshold
Time dependence of activity from neutron capture is based on exposure and decay. Constant rate of fluence F Minimal loss of target NT Typical Problem A 3-g sample of 32S is irradiated with fast neutrons at 155 cm-2s-1. The cross section is 0.200 barn. What is the maximum activity? Answer The number of target nuclei, The maximum is for large t. Neutron Activation