Chapter 11 Trace Elements: Metals, Paints and Soil
170 likes | 1.07k Views
Chapter 11 Trace Elements: Metals, Paints and Soil. Objectives: Describe the usefulness of trace elements for the forensic comparison of various types of physical evidence. Distinguish between a continuous and line emission spectrum. Describe the parts of a simple emission spectrograph.

Chapter 11 Trace Elements: Metals, Paints and Soil
E N D
Presentation Transcript
Chapter 11 Trace Elements: Metals, Paints and Soil Objectives: Describe the usefulness of trace elements for the forensic comparison of various types of physical evidence. Distinguish between a continuous and line emission spectrum. Describe the parts of a simple emission spectrograph. Define protons, neutrons, and electrons, including their mass and charge relationships. Define atomic number and atomic mass number. Describe the orbital energy levels that are occupied by electrons. State what happens when an atom absorbs a definite amount of energy. Explain the phenomenon of an atom releasing energy in the form of light. Define an isotope. Define radioactivity. Explain how elements can be made radioactive.
Chapter 11 Trace Elements: Metals, Paints and Soil Objectives: Describe the components of paint. Classify automobile paints. List those examinations most useful for performing a forensic comparison of paint. Describe the proper collection and preservation of paint evidence. List the important forensic properties of soil. Describe the density-gradient tube technique. Describe the proper collection of soil evidence.
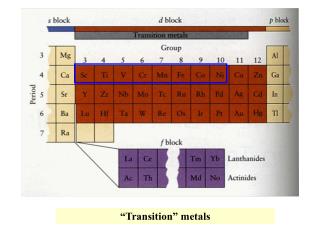
Forensic Analysis of Metals • Trace Elements • small quantities of elements found as impurities in all natural and manufactured products • useful as “invisible”markers • Kennedy Assassination • bullets and fragments tested • for silver and antimony • match fragments in car • Connally bullets indistinguishable • Q1 and Q1 same Ag Sb ppm • Kennedy bullets indistinguishable • Q2 and Q14 • Connally and Kennedy bullets are different
Atomic Structure • Atoms composed of particles: • Proton • positive charge • mass of 1 • Neutron • no charge • mass of 1 • Electron • negative charge • mass of 1/1837 • Proton number distinguishes elements • Atomic Number = # of protons • Mass Number = sum of protons & neutrons nucleus
Elements and Isotopes • Isotope • Atoms of same element • same atomic number (electrons & protons) • Have different atomic mass • different number of neutrons • Radioisotope • unstable isotope • decompose
Isotopes • atoms of same element with different masses • same # of protons • different # of neutrons
Radioactivity • high-energy particles released during decay • Alpha rays • largest/least energy • He nucleus (2 protons & 2 neutrons) • Beta Rays • smaller/more energy • electrons • Gamma Rays • smallest/most energy • electromagnetic radiation
Neutron Activation Analysis • atoms in a nuclear reactor gain neutrons • becoming unstable • releasing gamma rays • can be detected to identify trace element • determine if samples have common origin
Composition of Paint • pigments • impart color and opacity • organic compounds • binder • polymer and solvent • polymer holds pigments • solvent evaporates after application
Automotive Paints • Electrocoat Primer • organic resins electroplated to resist erosion (black or gray) • Primer Surfacer • epoxy modified polyester or urethane • smooth out and hide seams • Basecoat • acrylic polymer • pigments • mica chips aluminum flakes • resist weather, UV, acid rain • Clearcoat • acrylic or polyurethane • improve gloss
Microscopic Paint Examination • side by side comparison • layer structure • match # and sequence of colors • “chip” fit
Paint Binder Characterization • Pyrolysis Gas Chromatography • decompose paint sample w/heat • pyrolyze • expose to high temperatures (500-1000°C) • sample breaks up into many gaseous parts • run these gases through GC • forms a pyrogram • can distinguish polymers • Infrared Spectrophotometry • each “binder” will have a characteristic absorption spectra
Paint Pigment Characterization • Inorganic Pigments • identified by emission spectra, neutron activation analysis, x-ray spectroscopy • detects 15 to 20 elements • most are common to all paints (unimportant) • a few are rarely found (important) • Organic Pigments • UV Absorption Spectra • Carbon Arc Emission Spectrometry • “fingerprint” from “excited” electrons • absorb energy move from ground state to excited state • release “light” energy when they return to ground state • Inductively Coupled Emission Spectrometry (ICP) • argon gas and radio frequency induced coil form “hot plasma” • aerosol of sample forced through the plasma forming an emission spectra
Significance of Paint Evidence • Canadian Study • 260 vehicle chips in wreck yard • color, layer, and IR spec study • all but one pair were distinguishable • PDQ (Paint Data Query) • Canadian data base started 1975 • automotive paints • layers, primer color, binder
Collection of Paint Evidence • primary goal • keep chip intact • druggist fold or vial • clothing • package “whole” • hit and runs • get standard sample from undamaged area • scrape to bare metal • ¼ inch2 sample sufficient
Forensic Examination of Soil • Visual Comparison • color and texture • soils compared must be dried under identical lab conditions • Microscopic Comparison • low power • plant, animal, artificial materials • high power • minerals and rocks (geologist) • Density Tube Gradient • tubes with liquids of various densities • density distribution pattern of soils • useful only in conjunction with other tests
Collection of Soil Evidence • must establish variation at scene • standards collected at various intervals within 100 yard radius • only surface soil • each in individual plastic vials • suspect samples • take whole object containing soil • car soil • “lump form” sampling • soil laid down in layers • unique layering may have mre value