Ionic Bonds
Ionic Bonds. Building Science Champions. Objectives. Explain the differences between an atom and an ion. Describe how an ionic bond forms. Identify the properties of ionic compounds. Key terms. Ion Ionic bonds Polyatomic ion Crystal . Electron Transfer.

Ionic Bonds
E N D
Presentation Transcript
Ionic Bonds Building Science Champions
Objectives • Explain the differences between an atom and an ion. • Describe how an ionic bond forms. • Identify the properties of ionic compounds.
Key terms • Ion • Ionic bonds • Polyatomic ion • Crystal
Electron Transfer • In order for elements to bond with one another they must have an open place for a valence electron. • Atoms that have 4 or fewer valence electrons are said to lose electrons. • Atoms that have 5 or more valence electrons are said to gain electrons.
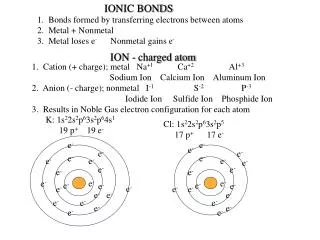
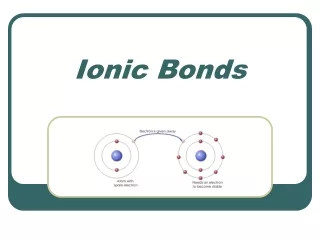
Ion • An ion is an atom or group of atoms that have become electrically charged. • Atoms become electrically charged by gaining or losing an electron. • When an atom loses an electron it loses a negative charge and becomes positive. • When an atom gains an electron, it gains a negative charge and becomes negative.
Ions and their charges A plus charge represent elements/compounds that have lost an electron, a negative charge represents elements/compounds that have gained an electron. A subscript represents the number of that element only.
Bonds,Ionic bonds! • Anionic bond is the attraction between two oppositely charged ions. (Usually between metals and nonmetals) • When the two ions come together, the opposite charges cancel each other out. • Locate Sodium on your ion chart. What is its charge? • Locate Chloride on your ion chart. What is its charge? Na + Cl Na+Cl-
Ionic Bonds • Compounds are electrically neutral. • Ions come together to balance out each others charge. • The NaCl example is a 1:1 ratio, meaning there is 1 Sodium ion and 1 Chlorine ion. • How many chloride ions would be needed to cancel out magnesium? 2 chloride ions
Ionic Bonds • Reaction between metals and nonmetals often result in IONIC COMPOUNDS. • These reactions easily occur between the metals in Group 1 and halogens in Group 17. • Why do you think these two families easily bond? Their number of valence electrons. Trying to get 8 valence electrons and by adding them together the magic number of 8 is achieved.
Ionic Questions? • How could an atom of sodium become more stable? Lose an electron • How could chlorine become more stable? Gain an electron • What happens when sodium and chlorine bond? They neutralize each other
Polyatomic Ions • Polyatomic ions are made of more than one atom. • Polyatomic ions are a group of atoms that work as one. • They have a positive or negative charge. • If a polyatomic ion combines with an ion of an opposite charge an ionic compound is formed. • Examples: CO32- = carbonate ion Ca2+ = calcium ion CaCO3 = calcium carbonate
Naming ion compounds • The name of the positive ion comes first, followed by the name of the negative ion. • The positive ion is usually a metal. • If a negative ion is in the compound, the end of its name changes to - ideMgO (magnesium oxide) • If the negative ion is polyatomic, its name is unchanged. Na2CO3 (sodium carbonate)
Properties of Ionic Compounds • Properties of ionic compounds include: • Crystal shape • High melting point • Electrical conductivity
CrystalShape • Ions form an orderly, three-dimensional arrangement called a crystal. • In ionic compounds, every ion is attracted to ions near it that have an opposite charge • Positive ions near negative ions and negative ions near positive ions.
High melting points • Ions are held together in a crystal by attractions between oppositely charged particles. • Ions separate when the particles have enough energy to break apart. • It takes temperature of 801OC
Electrical conductivity • When ionic compounds are placed in water, the solution conducts electricity. • Electricity is the flow of electric charge and ions have electric charge. • A solid ionic compound does not mix electricity well. • Melting ionic compounds also allows them to conduct electricity.
Review • An ion is an atom or group of atoms that have become electrically charged. • Ions are either positive or negative. • An ionic bond is the attraction between 2 oppositely charged ions. • Compounds are electrically neutral. • When atoms have 4 or fewer electrons, they will LOSE electrons and atoms with 5 or greater electrons will GAIN electrons. This process takes place during bonding.
References • Anderson, M. et all (2012) Physical Science. McGraw-Hill: Columbus • Frank, D.V et al (2001). Physical Science. Prentice Hall: New Jersey