Basic Gas Chromatography
Basic Gas Chromatography. Prepared by: Mina S. Buenafe. Gas Chromatography Chromatography – A Very Brief History Definitions / Terminologies in GC Instrumentation Overview System Modules Mobile Phase (Carrier Gas) Inlets Stationary Phase(s) Columns (Packed and Capillary) Detector(s)

Basic Gas Chromatography
E N D
Presentation Transcript
Basic Gas Chromatography Prepared by: Mina S. Buenafe
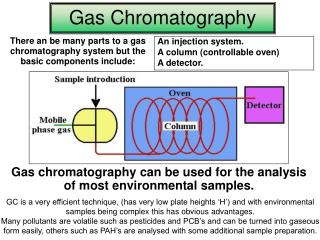
Gas Chromatography • Chromatography – A Very Brief History • Definitions / Terminologies in GC • Instrumentation Overview • System Modules • Mobile Phase (Carrier Gas) • Inlets • Stationary Phase(s) • Columns (Packed and Capillary) • Detector(s) • Troubleshooting
Chromatography – A (Very) Brief History IN THE EARLY 1900’S M. Tswett published his work on separation of plant pigments. He coined the term chromatography (literally translated as color writing) and scientifically described the process – earning him the title “Father of Chromatography” W. Ramsey published his work on separation of mixture of gases and vapors on adsorbents like charcoal. IN THE EARLY 1940’s A. Martin and R. Synge first suggested the possibilities of gas chromatography in a paper published in Biochem. J., v.35, 1358, (1941). Martin won a Nobel Prize for his work in Partition chromatography. IN THE EARLY 1950’s A. Martin and A. James published the epic paper describing the first gas chromatograph
Definition of Terms Chromatography: A physical method of separation in which the components to be separated are distributed between two phases, one of which is stationary while the other moves in a definite direction “Official” IUPAC definition
Definition of Terms Chromatogram It is the output signal from the detector of the instrument.
Definition of Terms Distribution Constant (KC) It is the tendency of a given component to be attracted to the stationary phase. This can be expressed in chemical terms as an equilibrium constant. Also called the partition coefficient (KP) or the distribution coefficient (KD) KC = [A]S/[A]M Mathematically, it is defined as the concentration of solute A in the stationary phase divided by its concentration in the mobile phase.
Definition of Terms The attraction to the stationary phase can also be classified according to the type of sorption by the solute. Adsorption: sorption on the surface of the stationary phase Absorption: sorption into the bulk of the stationary phase (usually called ‘partition’ by chromatographers)
Definition of Terms Retention Volume (VR) It is usually defined as the distance between the point of injection to the peak maximum. It is the volume of the carrier gas necessary to elute the solute of interest. Mathematically: VR = FC x tR Where FC is the constant flow rate tR is the retention time
Definition of Terms Phase Ratio (b) For packed columns: • b = Mobile Phase Volume • Stationary Phase Volume • For capillary columns: • b = rc/2df • Where rc is the radius of the column • df is the thickness of the film
Definition of Terms Retention Factor (k) It is the ratio of the amount of the solute (NOT concentration) in the stationary phase to the amount in the mobile phase. It is also called capacity factor (k’), capacity ratio, or partition ratio Mathematically: k = (WA)S/(WA)M = KC/b Also k = (tR - t0) = time in stationary phase t0 time in carrier gas k is temperature and flow dependent. Best separations occur when k is between 5 and 7
Definition of Terms Theoretical Plates (N) This is the most common measure of column efficiency in chromatography N = 16(tR/Wb)2 = 5.54(tR/ Wh)2 Where Wb is thepeak width at the base Wh is the peak width at half-height
Definition of Terms Height Equivalent to a Theoretical Plate (H) This is a related parameter that also defines column efficiency. Also identified as HETP Mathematically: H = L/N Where L is the Column Length (An efficient column will have a large N and a small H)
Definition of Terms Separation Factor (a) It is a measure of relative distribution constants. Also known as selectivity and/or solvent efficiency. Mathematically: a = k2/k1 = (KC)2/(KC)1 It is dependent on: • Chemical composition of the phase • Partitioning between the two phases
Definition of Terms Resolution (Rs) It is the degree to which adjacent peaks are separated. Mathematically: Rs = (tR)B – (tR)A [(Wb)B + (Wb)A]/2 Also Rs =—L/H x k/(k+1) x a-1/a
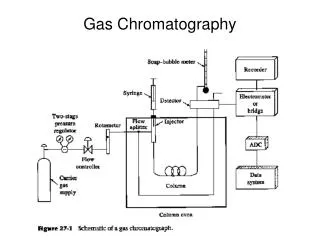
Instrumentation Overview Schematic of a Typical GasChromatograph
System Modules Main purpose: carries the sample through the column Carrier Gas Secondary purpose: provides a suitable matrix for the detector to measure the sample component. Carrier gases should be of high purity (minimum of 99.995%). • Oxygen & water impurities can chemically attack the liquid phase of the column and destroy it. • Trace water content can desorb other column contaminants and produce high detector background or ‘ghost peaks’. • Trace hydrocarbon contents can cause high detector background with FID’s and limit detectability.
System Modules Flow Measurements and Control: • Essential for column efficiency and qualitative analysis (e.g. reproducibility of retention times) Average linear flow velocity (ū) in OT columns: ū = L/tm where L is column length in cm tm is the retention time of an unretained peak (e.g. methane) in sec To convert linear flow velocity to flow rate (Fc) in mL/min: Fc = ū x Pr2 x 60sec/min Carrier Gas
System Modules Carrier Gas Effect of mobile phase (carrier gas) density on column efficiency. Van Deemter plots for the 3 common carrier gases for a column of capacity factor k’ = 7.90. The low density gases (H2 & He) have optimum efficiency at slightly higher flow rates than N2. The much lower slopes of H2 and He curves allow them to be used at higher flow rates (compared to N2) with very little loss of separation efficiency.
System Modules Inlets Inlets are the points of sample introduction Ideal Sample Inlets for Column Type:
System Modules Inlets Split Injector The oldest, simplest, and easiest injection technique. Cross section of a typical split injector Advantages to Split Injection: • High resolution separations • Neat samples can be introduced. • Dirty samples can be introduced by putting a deactivated glass wool plug in the liner to trap non-volatile components Disadvantages: • Trace analysis is limited • Process sometimes discriminates between high molecular weight solutes so that the sample entering the column is not representative of the sample injected.
System Modules Inlets Splitless Injector Samples have to be diluted in a volatile solvent and 1-5mL is injected in the heated injection port. Septum purge is essential in splitless injections. Cross section of a typical splitless injector Advantages to Splitless Injection: • Improved sensitivity over a split injector Disadvantages: • Time consuming • Initial temperature and time of opening the split valve needs to be optimized. • Not well suited for volatile compounds (boiling points of peaks of interest have to be about 30oC higher than solvent.
System Modules Inlets Other Types of Inlets: • Direct Injection: involves injecting a small sample into a glass liner where vapors are carried directly into the column. • On-Column Injection: inserting the precisely aligned needle into the capillary column and making injections inside the column. • Flash Vaporization: involves heating the injection port to a temperature well above the boiling point to ensure rapid volatilization • Static Headspace: concentrates the vapors over a solid or liquid sample (best for residual solvent analysis)
System Modules Stationary Phase Sub-classification of GC Techniques • GSC: gas solid chromatography - stationary phase is solid • GLC: gas liquid chromatography -stationary phase is liquid
System Modules Gas Solid Chromatography (GSC) • Solids used are traditionally run in packed columns • These solids should have small and uniform particle sizes (e.g. 80/100 mesh range) Stationary Phase Some of these solids have been coated on the inside walls of capillary columns and are called “Support Coated Open Tubular” or SCOT columns.
System Modules Stationary Phase One major application of Packed Column GSC is in Gas Analysis. Reasons: • Adsorbents provide high surface areas for maximum interaction with gases that may be difficult to retain on liquid stationary phase. • Large samples can be accommodated, providing lower absolute detection limits. • Some packed column GC’s can be configured to run below ambient temperature which will also increase the retention of the gaseous solutes. • Unique combinations of multiple columns and/or valving make it possible to optimize analysis of a particular sample. Packed Columns also provide the flexibility of allowing mixed packings for special applications (e.g. 5% Fluorcol on Carbopack B® for analysis of Freons)
System Modules Stationary Phase Gas Liquid Chromatography (GLC) To use liquid as stationary phase,techniques were applied to hold the liquid in a column. • For packed columns: liquid is coated onto a solid support, chosen for its high surface area and inertness. The coated support is then dry-packed into a column as tightly as possible. • For capillary or open tubular (OT) columns: liquid is coated on the inside of the capillary. To make it adhere better, the liquid phase is often extensively cross-linked and sometimes chemically bonded to the fused silica surface. Schematic representation of (a) packed column and (b) capillary column
System Modules Stationary Phase Gas Liquid Chromatography (GLC) Requirements for the stationary liquid phase: • Low vapor pressure • Thermal stability • (if possible) Low viscosity (for fast mass transfer) • Should interact with the components of the sample to be analyzed (“Like dissolves like”)
System Modules Stationary Phase Gas LiquidChromatography (GLC) Types of Capillary Columns (OT) WCOT: Wall-coated open tubular column (provides the highest resolution of all OT’s – i.d.’s range from 0.1mm to 0.53mm and film thickness from 0.1 – 5.0m) PLOT: Porous layer open tubular column (less than 5% of all GC use these days) SCOT: Surface-coated open tubular column (no longer available in fused silica)
System Modules Detectors The part of the system that ‘senses’ the effluents from the column and provides a record of the analysis in the form of a chromatogram. The signals are proportional to the quantity of each analyte.
System Modules FID: Flame Ionization Detector The most common GC detector used. The column effluent is burned in a small oxy-hydrogen flame producing some ions in the process. These ions are collected and form a small current that becomes the signals. When no sample is being burned, there should be little ionization, the small current is produced from impurities from the from the hydrogen and air supplies. Hydrogen flow rate is commonly set to 40 – 45mL/min, Air, 350- 450mL/min, and for OT columns (with flows of about 1 mL/min), Make-Up gases is added to carrier gas (to make up the flow to 30mL/min) Detectors
System Modules Detectors TCD: Thermal Conductivity Detector This is a differential detector that measures the thermal conductivity of the analyte in the carrier gas compared to the thermal conductivity of the pure gas. At least two cavities are required. These cavities are drilled into a metal block and each contain a hot wire or filament. The filaments are incorporated into a Wheatstone Bridge Circuit (for resistance measurements). The choice of carrier gas will depend on the thermal conductivity of the analyte (H2 and He have highest TC’s, N2 gives rise to unusual peak shapes)
System Modules Detectors NPD: Nitrogen Phosphorus Detector A bead of Rb or Cs is electrically heated when flame ionization occurs. The detector shows enhanced detectability for nitrogen-, phosphorus-, or halogen- containing samples.
System Modules Detectors MSD: Mass Spectrometric Detector Analyte molecules are first ionized in order to be attracted or repelled by the proper magnetic or electrical fields.
Troubleshooting Common GC Problems: Retention Time Problems Resolution Problems Baseline Problems Peak Problems
Retention Time Problems Retention Time Shift
Retention Time Problems Retention Time Shift
Resolution Problems Decrease in separation Loss of Resolution
Resolution Problems Increase in peak width Loss of Resolution
Baseline Problems Excessive Column Bleed
Baseline Problems • Erratic Baseline (drift, wander)
Baseline Problems • Erratic Baseline (drift, wander)
Baseline Problems ·Noisy Baseline
Baseline Problems ·Noisy Baseline
Baseline Problems ·Ghost Peaks
Peak Problems ·Fronting Peaks
Peak Problems ·Tailing Peaks
Peak Problems ·Tailing Peaks
Peak Problems ·Tailing Peaks
Peak Problems ·Split Peaks
Peak Problems ·Split Peaks