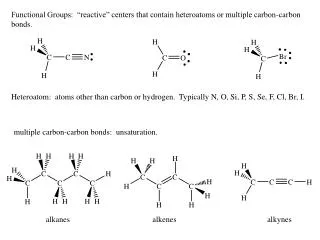
Understanding Alkenes: Bonding, Reactions, and Mechanisms in Organic Chemistry
This overview explores the bonding characteristics of alkenes, focusing on carbon's electron configuration changes and the formation of sp² hybrid orbitals. It explains the formation of sigma (σ) and pi (π) bonds through orbital overlap, and how unsaturation can be tested with bromine water. The description of reaction mechanisms includes electrophilic addition, carbocation formation, and reactions with hydrogen halides. Additionally, the processes for synthesizing alcohols and alkanes from alkenes via steam addition and hydrogenation are detailed.

Understanding Alkenes: Bonding, Reactions, and Mechanisms in Organic Chemistry
E N D
Presentation Transcript
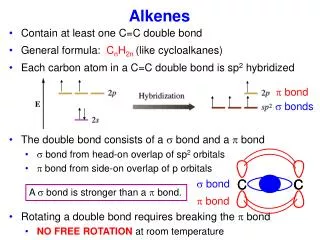
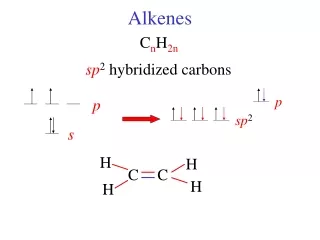
Bonding The electron configuration of carbon is 1s22s22p2 When carbon bonds in alkenes, the electron configuration changes 1s22s12p3 The 2s electron and two of the 2p electrons combine to form three sp2 hybrid orbitals, leaving a spare p-orbital on each of the carbon atoms
H H C C H A hybrid orbital from each carbon overlaps to form a σ-bond H
The p-orbitals overlap causing areas of electron density spanning across the top and the bottom of the σ-bond joining the two carbon atoms. This is the π-bond.
Test for unsaturation • If an alkene is shaken with bromine water (Br2) the colour changes from reddy-brown to colourless as dihalogenoalkanes are formed.
Reaction Mechanism • This is a two step reaction; • Formation of a carbocation by electrophillic addition • Rapid reaction with the negative Br ion
Reactions with hydrogen halides If an alkene is bubbled through concentrated HCl(aq) at room temperature, a monosubstituted chloro-alkane is formed.
Addition of steam When steam and a gaseous alkene are passed over a solid phosphoric acid catalyst (H3PO4) at 600K and 6MPa, alcohols are formed.
Addition of hydrogen If a gaseous alkene and H2 gas are passed over a finely divided nickel catalyst at high pressure and temperature (420K and 500kPa for making margarine!) an alkane is formed.