Understanding Reaction Mechanisms: Key Concepts and Examples
230 likes | 346 Views
Discover the importance of reaction mechanisms in chemical reactions, including elementary steps, molecularity, and rate-determining steps. Learn how to determine and evaluate reaction mechanisms using chemical intuition.

Understanding Reaction Mechanisms: Key Concepts and Examples
E N D
Presentation Transcript
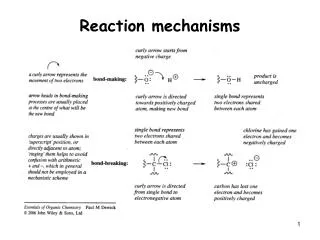
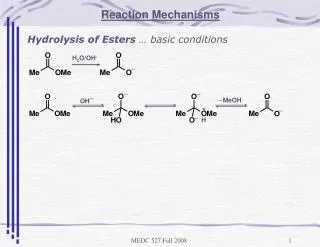
Rxn Mechanism • The series of steps that chemical reactions occur by • main points of kinetics is to learn these steps
NO2(g)+CO(g)-->NO(g)+CO2(g) • Rate = k[NO2]2 • Tells us about reactants and products and stoichiometry, but nothing about mechanism
NO2(g)+CO(g)-->NO(g)+CO2(g) • Mechanism thought to be: • NO2(g)+NO2(g)-->NO3(g)+ NO(g) • NO3(g)+CO(g)-->NO2(g)+CO2(g) • Two new rate constants k1, k2
NO3(g) ??? • INTERMEDIATE- species that is neither a reactant or a product, but is formed and used during the rxn sequence
Elementary Steps • Reactions whose rate law can be written from its molecularity
Molecularity • Number of species that must collide to produce the reaction indicated by that step
Unimolecular • Rxn involving one molecule • A-->prod • Rate = k[A]
Bimolecular • Involving two molecules • A + A --> prod • A + B --> prod • Rate = k[A]2 • Rate = k[A][B]
Termolecular • Involving 3 molecules • rare- hard for 3 to collide simultaneously • A + B + C --> prod • Rate = k[A][B][C]
Rxn mech- precisely • 1. The sum of elementary steps must give overall balanced eqn for rxn.
Rxn mech- precisely • 2. The mechanism must agree with the experimentally determined rate law
Question • Does our mechanism balance? • YES
Rate-determining step • The SLOW step in a rxn • Reactants can only become products as fast as their slow step will allow
Back to example • Assume that the first rxn is the SLOW step • Formation of NO3 occurs more slowly than its reaction with CO
Therefore... • The rate of CO2 production is controlled by the rate of formation of NO3
Since... • It is an elementary step, we can write the rate law from the molecularity • Rate = k[NO2]2
And look! • The overall rate can be no faster than the slowest step • Overall rate = k[NO2]2 !
Finding mechanism • Always find rate law first • Then use chemical intuition and try to make mechanism
But Remember • Mechanism can NEVER be proved absolutely
2NO2(g) + F2(g) 2NO2F(g) Rate Law = k[NO2][F2] Mechanism NO2 + F2 NO2F + F slow F + NO2 NO2F fast