Reaction mechanisms
Reaction mechanisms. Ionic Reactions. Ionic Reactions. Ionic Reactions. Ionic Reactions. Bond Polarity. Partial charges. Nucleophiles and Electrophiles. Leaving Groups. Radical Reactions. Type of Reactions. Nucleophilic reactions: nucleophilic substitution (S N ).

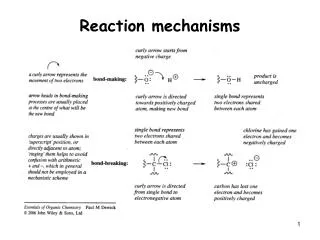
Reaction mechanisms
E N D
Presentation Transcript
Bond Polarity Partial charges
Nucleophilic reactions: nucleophilic substitution (SN) • in the following general reaction, substitution takes place on an sp3 hybridized (tetrahedral) carbon Nucleophilic substitution: -> reagent is nucleophil -> nucleophil replaces leaving group -> competing reaction (elimination + rearrangements)
Nucleophilic Substitution • Some nucleophilic substitution reactions
Mechanism • Chemists propose two limiting mechanisms for nucleophilic displacement • a fundamental difference between them is the timing of bond breaking and bond forming steps • At one extreme, the two processes take place simultaneously; designated SN2 • S = substitution • N = nucleophilic • 2 = bimolecular (two species are involved in the rate-determining step) • rate = k[haloalkane][nucleophile] • In the other limiting mechanism, bond breaking between carbon and the leaving group is entirely completed before bond forming with the nucleophile begins. This mechanism is designated SN1 where • S = substitution • N = nucleophilic • 1 = unimolecular (only one species is involved in the rate-determining step) • rate = k[haloalkane]
SN2 reaction: bimolecular nucleophilic substitution • both reactants are involved in the transition state of the rate-determining step • the nucleophile attacks the reactive center from the side opposite the leaving group
SN2 • An energy diagram for an SN 2 reaction • there is one transition state and no reactive intermediate
SN1 reaction: unimolecular nucleophilic substitution • SN1 is illustrated by the solvolysis of tert-butyl bromide • Step 1: ionization of the C-X bond gives a carbocation intermediate
SN1 • Step 2: reaction of the carbocation (an electrophile) with methanol (a nucleophile) gives an oxonium ion • Step 3: proton transfer completes the reaction
SN1 • An energy diagram for an SN1 reaction
C H C H 6 5 6 5 + CH O C C OCH 3 3 H H Cl Cl (S)-Enantiomer (R)-Enantiomer A racemic mixture SN1 • For an SN1 reaction at a stereocenter, the product is a racemic mixture • the nucleophile attacks with equal probability from either face of the planar carbocation intermediate
Effect of variables on SN Reactions • the nature of substituents bonded to the atom attacked by nucleophile • the nature of the nucleophile • the nature of the leaving group • the solvent effect
Effect of substituents on SN reactions • SN1 reactions • governed by electronic factors, namely the relative stabilities of carbocation intermediates • relative rates: 3° > 2° > 1° > methyl • SN2 reactions • governed by steric factors, namely the relative ease of approach of the nucleophile to the site of reaction • relative rates: methyl > 1° > 2° > 3°
Effect of substituents on SN reactions • Effect of electronic and steric factors in competition between SN1 and SN2 reactions
Nucleophilicity • Nucleophilicity: a kinetic property measured by the rate at which a Nu attacks a reference compound under a standard set of experimental conditions • for example, the rate at which a set of nucleophiles displaces bromide ion from bromoethane • Two important features: • An anion is a better nucleophile than a uncharged conjugated acid • strong bases are good nucleophiles
The Leaving Group • the best leaving groups in this series are the halogens I-, Br-, and Cl- • OH-, RO-, and NH2- are such poor leaving groups that they are rarely if ever displaced in nucleophilic substitution reactions
Solvent Effect • Protic solvent: a solvent that contains an -OH group • these solvents favor SN1 reactions; the greater the polarity of the solvent, the easier it is to form carbocationsin it
Solvent Effect • Aprotic solvent: does not contain an -OH group • it is more difficult to form carbocations in aprotic solvents • aprotic solvents favor SN2 reactions
Competing Reaction: Elimination -Elimination: removal of atoms or groups of atoms from adjacent carbons to form a carbon-carbon double bond • we study a type ofb-eliminationcalled dehydrohalogenation (the elimination of HX)
b-Elimination • There are two limiting mechanisms for β-elimination reactions • E1 mechanism:at one extreme, breaking of the C-X bond is complete before reaction with base breaks the C-H bond • only R-X is involved in the rate-determining step • E2 mechanism:at the other extreme, breaking of the C-X and C-H bonds is concerted • both R-X and base are involved in the rate-determining step
E2 Mechanism • A one-step mechanism; all bond-breaking and bond-forming steps are concerted
E1 Mechanism • Step 1: ionization of C-X gives a carbocation intermediate • Step 2: proton transfer from the carbocation intermediate to a base (in this case, the solvent) gives the alkene Nucleophile -> acting as a strong base
Elimination • Saytzeff rule: the major product of a elimination is the more stable (the more highly substituted) alkene
Elimination Reactions • Summary of E1 versus E2 Reactions for Haloalkanes
Substitution vs Elimination • Many nucleophiles are also strong bases (OH- and RO-) and SN and E reactions often compete • the ratio of SN/E products depends on the relative rates of the two reactions • What favors Elimination reactions: • attacking nucleophil is a strong and large base • steric crowding in the substrate • High temperatures and low polarity of solvent
SN1 versus E1 • Reactions of 2° and 3° haloalkanes in polar protic solvents give mixtures of substitution and elimination products
SN2 versus E2 • It is considerably easier to predict the ratio of SN2 to E2 products
Summary of S vs E for Haloalkanes • for methyl and 1°haloalkanes
Summary of S vs E for Haloalkanes • for 2° and 3° haloalkanes
Summary of S vs E for Haloalkanes • Examples: predict the major product and the mechanism for each reaction Elimination, strong base, high temp. SN2, weak base, good nucleophil SN1 (+Elimination), strong base, good nucleophil, protic solvent No reaction, I is a weak base (SN2) I better leaving group than Cl
Carbocation rearrangements Also 1,3- and other shifts are possible The driving force of rearrangements is -> to form a more stable carbocation !!! Happens often with secondary carbocations -> more stable tertiary carbocation
Carbocation rearrangements in SN + E reactions Rearrangement
Carbocation rearrangements in SN + E reactions -> Wagner – Meerwein rearrangements Rearrangement of a secondary carbocations -> more stable tertiary carbocation Plays an important role in biosynthesis of molecules, i.e. Cholesterol -> (Biochemistry)
Carbocation rearrangements in Electrophilic addition reactions