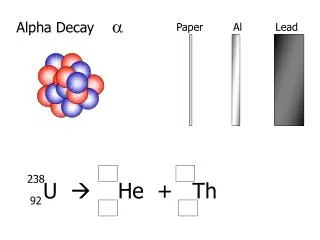
Alpha Decay
Alpha Decay. Readings Nuclear and Radiochemistry: Chapter 3 Modern Nuclear Chemistry: Chapter 7 Energetics of Alpha Decay Theory of Alpha Decay Hindrance Factors Heavy Particle Radioactivity Proton Radioactivity Identified at positively charged particle by Rutherford

Alpha Decay
E N D
Presentation Transcript
Alpha Decay • Readings • Nuclear and Radiochemistry: Chapter 3 • Modern Nuclear Chemistry: Chapter 7 • Energetics of Alpha Decay • Theory of Alpha Decay • Hindrance Factors • Heavy Particle Radioactivity • Proton Radioactivity • Identified at positively charged particle by Rutherford • Helium nucleus (4He2+) based on observed emission bands • Energetics • Alpha decay energies 4-9 MeV • Originally thought to be monoenergetic, fine structure discovered • AZ(A-4)(Z-2) + 4He + Qa
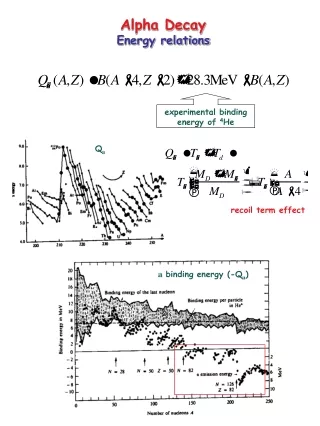
Alpha Decay Energetics • Q value positive for alpha decay • Q value exceeds alpha decay energy • maTa = mdTd • md and Td represent daughter • From semiempirical mass equation • emission of an α-particle lowers Coulomb energy of nucleus • increases stability of heavy nuclei while not affecting overall binding energy per nucleon • tightly bound α-particle has approximately same binding energy/nucleon as original nucleus • Emitted particle must have reasonable energy/nucleon • Energetic reason for alpha rather than proton • Energies of alpha particles generally increase with atomic number of parent
Energetics • Calculation of Q value from mass excess • 238U234Th + a + Q • Isotope Δ (MeV) 238U 47.3070 234Th 40.612 4He 2.4249 • Qa=47.3070 – (40.612 + 2.4249) = 4.270 MeV • Q energy divided between α particle and heavy recoiling daughter • kinetic energy of alpha particle will be slightly less than Q value • Conservation of momentum in decay, daughter and alpha are equal rd=r • recoil momentum and -particle momentum are equal in magnitude and opposite in direction • p2=2mT where m= mass and T=kinetic energy • 238Ualpha decay energy
Energetics • Kinetic energy of emitted particle is less than Coulomb barrier α-particle and daughter nucleus • Equation specific of alpha • Particles touching • For 238 U decay • Alpha decay energies are small compared to required energy for reverse reaction • Alpha particle carries as much energy as possible from Q value, • For even-even nuclei, alpha decay leads to ground state of daughter nucleus • as little angular momentum as possible • ground state spins of even-even parents, daughters and alpha particle are l=0
Alpha decay theory • Distance of closest approach for scattering of a 4.2 MeV alpha particle is ~62 fm • Distance at which alpha particle stops moving towards daughter • Repulsion from Coulomb barrier • Alpha particle should not get near nucleus • should be trapped behind a potential energy barrier • Wave functions are only completely confined by infinitely highpotentialenergy barriers • With finite size barrier wave function has different behavior • main component inside barrier • finite piece outside barrier • Tunneling • trapped particle has component of wave function outside potential barrier • Some probability to go through barrier • Related to decay probability • Higher energy has higher tunneling probability Vc Alpha decay energy
Alpha Decay Theory • Closer particle energy to barrier maximum more likely particle will penetrate barrier • More energetic alpha will encounter barrier more often • Increase probability of barrier penetration due to • Geiger Nuttall law of alpha decay • constants A and B have Z dependence. • simple relationship describes data on α-decay • over 20 orders of magnitude in decay constant or half-life • 1 MeV change in -decay energy results in a change of 105 in half-life
Alpha Decay Calculations • Alpha particle barrier penetration from Gamow • T=e-2G • Determination of decay constant from potential information • Using square-well potential, integrating and substituting • Z daughter, z alpha
Gamow calculations • From Gamow • Calculated emission rate typically one order of magnitude larger than observed rate • observed half-lives are longer than predicted • Observation suggest a route to evaluate alpha particle pre-formation factor
Alpha Decay Theory • Even-even nuclei undergoing l=0 decay • average preformation factor is ~ 10-2 • neglects effects of angular momentum • Assumes α-particle carries off no orbital angular momentum (ℓ = 0) • If α decay takes place to or from excited state some angular momentum may be carried off by α-particle • Results in change in decay constant when compared to calculated
Hindered -Decay • Previous derivation only holds for even-even nuclei • odd-odd, even-odd, and odd-even nuclei have longer half-lives than predicted due to hindrance factors • Assumes existence of pre-formed -particles • Ground-state transition from nucleus containing odd nucleon in highest filled state can take place only if that nucleon becomes part of -particle • therefore another nucleon pair is broken • less favorable situation than formation of an -particle from already existing pairs in an even-even nucleus • may give rise to observed hindrance • -particle is assembled from existing pairs in such a nucleus, product nucleus will be in an excited state • this may explain higher probability transitions to excited states • Hindrance from difference between calculation and measured half-life • Hindrance factors between 1 and 3E4 • Hindrance factors determine by • ratio of measured alpha decay half life over calculated alpha decay half life • ratio of calculated alpha decay constant over measured alpha decay constant
Hindrance Factors • Transition of 241Am (5/2-) to 237Np • states of 237Np (5/2+) ground state and (7/2+) 1st excited state have hindrance factors of about 500 (red circle) • Main transition to 60 keV above ground state is 5/2-, almost unhindered
Hindrance Factors • 5 classes of hindrance factors based on hindrance values • Between 1 and 4, transition is called a “favored” • emitted alpha particle is assembled from two low lying pairs of nucleons in parent nucleus, leaving odd nucleon in its initial orbital • Hindrance factor of 4-10 indicates a mixing or favorable overlap between initial and final nuclear states involved in transition • Factors of 10-100 indicate that spin projections of initial and final states are parallel, but wave function overlap is not favorable • Factors of 100-1000 indicate transitions with a change in parity but with projections of initial and final states being parallel • Hindrance factors of >1000 indicate that transition involves a parity change and a spin flip
Topic Review • Understand and utilize systematics and energetics involved in alpha decay • Calculate Q values for alpha decay • Relate to alpha energy and fine structure • Correlate Q value and half-life • Models for alpha decay constant • Tunneling and potentials • Hindered of alpha decay • Understand proton and other charged particle emission
Homework Questions • Calculate alpha decay Q value and Coulomb barrier potential for following, compare values • 212Bi, 210Po, 238Pu, 239Pu, 240Am, 241Am • What is basis for daughter recoil during alpha decay? • What is relationship between Qa and alpha decay energy (Ta) • What are some general trends observed in alpha decay? • Compare calculated and experimental alpha decay half life for following isotopes • 238Pu, 239Pu, 241Pu, 245Pu • Determine hindrance values for odd A Pu isotopes above • What are hindrance factor trends? • How would one predict half-life of an alpha decay from experimental data?
Pop Quiz • Calculate alpha decay energy for 252Cf and 254Cf from mass excess data below. • Which is expected to have shorter alpha decay half-life and why? • Calculate alpha decay half-life for 252Cf and 254Cf from data below. (use % alpha decay)
Beta Decay • Readings: Nuclear and Radiochemistry: Chapter 3, Modern Nuclear Chemistry: Chapter 8 • Neutrino Hypothesis • Derivation of Spectral Shape • Kurie Plots • Beta Decay Rate Constant • Selection Rules • Transitions • Majority of radioactive nuclei are outside range of alpha decay • Beta decay • Second particle found from U decay • Negative particle • Distribution of energies • Need another particle to balance spin • Parent, daughter, and electron • Need to account for half integer spin • Beta decay half-life • few milliseconds to ~ 1016 years • How does this compare to alpha decay?
-Decay • Class includes any radioactive decay process in which A remains unchanged, but Z changes • - decay, electron capture, + decay • energetic conditions for decay: • - decay: MZ MZ+1 • Electron capture: MZMZ-1, • + decay: MZ MZ-1+2me • + decay needs to exceed 1.02 MeV • Below 1.02 MeV EC dominates • + increases with increasing energy • Decay energies of -unstable nuclei rather systematically with distance from stability • Predicted by mass parabolas • Energy-lifetime relations are not nearly so simple as alpha decay • -decay half lives depend strongly on spin and parity changes as well as energy • For odd A, one -stable nuclide; for even A, at most three -stable nuclides • Information available from mass parabolas • Odd-odd nuclei near the stability valley (e.g., 64Cu) can decay in both directions • Form even-even nuclei • Beta particle energy not discrete • Continuous energy to maximum
The Neutrino • Solved problems associated with -decay • Continuum of electron emission energies • Zero charge • neutron -> proton + electron • Small mass • Electron goes up to Q value • Anti-particle • Account for creation of electron particle • spin of ½ and obeys Fermi statistics • couple the total final angular momentum to initial spin of ½ ħ, • np+ + e- is not spin balanced, need another fermion
Spin in Beta Decay • Spins of created particles can be combined in two ways • Electron and neutrino spin both 1/2 • S=1 in a parallel alignment • S= 0 in an anti-parallel alignment • two possible relative alignments of "created" spins • Fermi (F) (S=0) • Low A • Gamow-Teller (GT) (S =1) • High A • Spin change since neutron number tends to be larger than proton • A source can produce a mixture of F and GT spins
Q value calculation (Review) Beta decay • Find Q value for the Beta decay of 24Na • 1 amu = 931.5 MeV • M (24Na)-M(24Mg) • 23.990962782-23.985041699 • 0.005921 amu • 5.5154 MeV • From mass excess • -8.4181 - -13.9336 • 5.5155 MeV • Q value for the EC of 22Na • M (22Na)-M(22Ne) • 21.994436425-21.991385113 • 0.003051 amu • 2.842297 MeV • From mass excess • -5.1824 - -8.0247 • 2.8432 MeV • Q- are ~0.5 – 2 MeV, Q + ~2-4 MeV and QEC ~ 0.2 – 2 MeV • What about positron capture instead of EC? Positron decay Electron Capture
Positrons • Postulated in 1931 • Relativistic equations could be solved for electrons with positive energy states • Require energies greater than electron mass • Creation of positive hole with electron properties • Pair production process involves creation of a positron-electron pair by a photon in nuclear field • Nucleus carries off some momentum and energy • Positron-electron annihilation • Interaction of electron into a whole in sea of electrons of negative energy • simultaneous emission of corresponding amount of energy in form of radiation • Responsible for short lifetime of positrons • No positron capture decay • Annihilation radiation • energy carried off by two quanta of opposite momentum • Annihilation conserves momentum • Exploited in Positron Emission Tomography
P(pe)dpe probability electron with momentum pe+dpe e electron wave function n neutrino wave function e(0)2 and n(0)2 probability of finding electron and neutrino at nucleus Mifmatrix element characterizes transition from initial to final nuclear state Mif2 a measure of overlap amount between wave functions of initial and final nuclear states dn/dEo is density of final states with electron in specified momentum interval number of states of final system per unit decay energy Fermi constant (g) governs other interactions in addition to beta decay m-meson decay, p-meson decay, neutrino-electron scattering Weak interactions Weak Interaction
Weak Interaction • Integration over all electron momenta from zero to maximum should provide transition probabilities or lifetimes • Variations in number of electrons at a given energy • Derivation of emission spectrum • Classically allowed transitions both have electron and neutrino emitted with zero orbital angular momentum • Allowed have s orbital angular momentum • Relatively high probabilities for location of electron and neutrino at nucleas for s wave compared to higher l • p,d,f, etc. • 2 of allowed transitions 2of forbidden transitions • Magnitudes of (0) and Mif are independent of energy division between electron and neutrino
Weak Interaction • Spectrum shape determined entirely by e(0) and dn/dEo • dn/dEo density of final states with electron momentum • Coulomb interaction between nucleus and emitted electron (e(0)) neglected • Reasonable for low Z • Density of final states determined from total energy W • W is total (kinetic plus rest) electron energy • Wo is maximum W value • dn/dEo goes to zero at W = 1 and W = Wo • Yields characteristic bell shape beta spectra
Coulomb Correction • Agreement of experiment and modeling at low Z • At higher Z need a correction factor to account for coulomb interaction • Coulomb interaction between nucleus and emitted electron • decelerate electrons and accelerate positrons • Electron spectra has more low-energy particles • Positron spectra has fewer low-energy particles • Treat as perturbation on electron wave function e(0) • Called Fermi function • Defined as ratio of e(0)2Coul /e(0)2free • perturbation on e(0) and spectrum multiplied by Fermi function • Z daughter nucleus • v beta velocity • + for electrons • - for positron
Kurie Plot • Comparison of theory and experiment for momentum measurements • Square root of number of beta particles within a certain range divided by Fermi function plotted against beta-particle energy (W) • x axis intercept is Q value • Linear relationship designates allowed transition
Fermi Golden Rule • Used for transition probability • Treat beta decay as transition that depends upon strength of coupling between initial and final states • Decay constant given by Fermi's Golden Rule • matrix element couples initial and final states • density of states that are available to system after transition • Wave function of initial and final state • Operator which coupled initial and final state • Rate proportional to strength of coupling between initial and final states factored by density of final states available to system • final state can be composed of several states with the same energy • Degenerate states
Comparative Half Lives • Based on probability of electron energy emission coupled with spectrum and Coulomb correction fot1/2 • comparative half life of a transition • Assumes matrix element is independent of energy • true for allowed transitions • Yields ft (or fot1/2), comparative half-life • may be thought of as half life corrected for differences in Z and W • W is total kinetic energy • fo can be determine when Fermi function is 1 (low Z) • Rapid estimation connecting ft and energy • Simplified route to determine ft (comparative half-life)
Comparative half-lives • Z is daughter and Eo is maximum energy in MeV (Q value) • Log ft = log f + log t1/2 • t1/2 in seconds • 14 O to 14N • positron decay • Q=1.81 MeV • T1/2 =70.6 s • Log fb+ = 1.83, log t = 1.84 • Log ft=3.67
Log ft calculation • 212Bi beta decay • Q = 2.254 MeV • T1/2 = 3600 seconds • 64 % beta branch • lb=1.22E-4 s-1 • T1/2Beta =5625 seconds • Log f=3.73; log t=3.75 • Log ft=7.48
Log ft data • What drives changes in log ft values for 24Na and 205Hg? • Examine spin and parity changes between parent and daughter state
Extranuclear Effects of EC • If K-shell vacancy is filled by L electron, difference in binding energies emitted as x-ray or used in internal photoelectric process • Auger electrons are additional extranuclear electrons from atomic shells emitted with kinetic energy equal to characteristic x-ray energy minus its binding energy • Fluorescence yield is fraction of vacancies in shell that is filled with accompanying x-ray emission • important in measuring disintegration rates of EC nuclides • radiations most frequently detected are x-rays
Selection Rules • Allowed transitions are ones in which electron and neutrino carry away no orbital angular momentum • largest transition probability for given energy release • If electron and neutrino do not carry off angular momentum, spins of initial and final nucleus differ by no more than h/2 and parities must be same • 0 or 1 • Fermi or Gamow-Teller transitions • If electron and neutrino emitted with intrinsic spins antiparallel, nuclear spin change (I )is zero • singlet • If electron and neutrino spins are parallel, I may be +1, 0, -1 • triplet
All transitions between states of I=0 or 1 with no change in parity have allowed spectrum shape I is nuclear spin Not all these transitions have similar fot values transitions with low fot values are “favored” or “superallowed” emitters of low Z between mirror nuclei one contains n neutrons and n+1 protons, other n+1 neutrons and n protons Assumption of approximately equal Mif2 values for all transitions with I=0, 1 without parity change was erroneous Selection Rules
Forbidden Transitions • When transition from initial to final nucleus cannot take place by emission of s-wave electron and neutrino • orbital angular momenta other than zero • l value associated with given transition deduced from indirect evidence • ft values, spectrum shapes • If l is odd, initial and final nucleus have opposite parities • If l is even, parities must be same • Emission of electron and nucleus in singlet state requires I l • Triple-state emission allows I l+1
Other Beta Decay • Double beta decay • Very long half-life • 130Te and 82Se as examples • Can occur through beta stable isotope • 76Ge to 76Se by double beta • 76Ge to 76As • Q= -73.2130- (-72.2895) • Q= -0.9235 MeV • Possible to have neutrinoless double beta decay • two neutrinos annihilate each other • Neutrino absorbed by nucleon • Beta delayed decay • Nuclei far from stability can populate unbound states and lead to direct nucleon emission • First recognized during fission • 1 % of neutrons delayed • 87Br is produced in nuclear fission and decays to 87Kr • decay populates some high energy states in Kr daughter • 51 neutrons, neutron emission to form 86Kr
Topic Review • Fundamentals of beta decay • Electron, positron, electron capture • Neutrino Hypothesis • What are trends and data leading to neutrino hypothesis • Derivation of Spectral Shape • What influences shape • Particles, potentials • Kurie Plots • Beta Decay Rate Constant • Calculations • Selection rules • Log ft • How do values compare and relate to spin and parity • Other types of beta decay
Homework questions • For beta decay, what is the correlation between decay energy and half life? • What is the basis for the theory of the neutrino emission in beta decay. • In beta decay what are the two possible arrangements of spin? • What is the basis for the difference in positron and electron emission spectra? • What log ft value should we expect for the -decay to the 1- state of 144Pr? • Why is there no decay to the 2+ level? • Calculate and compare the logft values for EC, positron and electron decay for Sm isotopes.
Pop Quiz • Calculate the logft for the decay of 241Pu, 162Eu, 44Ti, and 45Ti. Provide the transition for each?