Alpha Decay basics
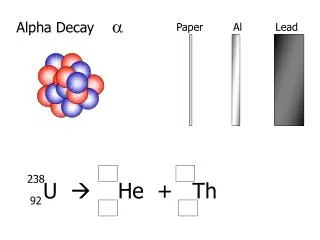
Alpha Decay basics. [Sec. 7.1/7.2/8.2/8.3 Dunlap]. Alpha decay Example Parent nucleus Cm-244. The daughter isotope is Pu-240. 96 Cm 244. 94 Pu 240. Why alpha particle instead of other light nuclei. Energy Q associated with the emission of various particles from a 235 U nucleus.

Alpha Decay basics
E N D
Presentation Transcript
Alpha Decaybasics [Sec. 7.1/7.2/8.2/8.3 Dunlap]
Alpha decay Example Parent nucleus Cm-244. The daughter isotope is Pu-240 96Cm244 94Pu240
Why alpha particle instead of other light nuclei Energy Q associated with the emission of various particles from a 235U nucleus.
There are always two questions that can be asked about any decay in atomic, nuclear or particle physics: (i) How much kinetic energy was released? and (ii) How quickly did it happen? (i.e. Energy? and Time?). Lets look at both of these questions for decay.
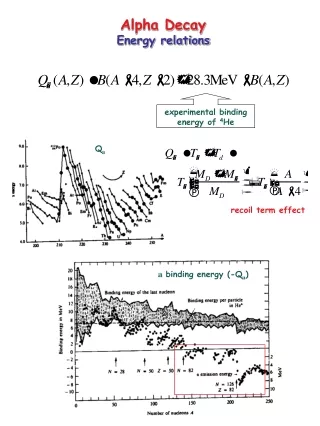
Energy Released Q Experiments The above diagram (right) shows the experimental energy of release. The above diagram (left) shows the abundance of alpha emitters. Both diagrams are as a function of A. Can you see the relationship?
The Energy of the α-particle, Tα Mass of X Q Mass of Y + particle And the energy released in the decay is simply given by energy
The Energy of the α-particle, Tα Conserving energy and momentum one finds: BEFORE AFTER +p, p2/8M -p, P2/2AM
Energy Released Q. This can be estimated from the SEMF by realizing that the B(Z,A) curve is rather smooth at large Z, and A and differential calculus can be used to calculate the B due to a change of 2 in Z and a change of 4 in A. Starting from (8.2) we also have:
There can be multiple alpha energies This diagram shows the alpha decay to the 240Pu daughter nucleus – and this nucleus is PROLATE and able to ROTATE collectively. Alpha decay can occur to any one of the excited states although not with the same probability. For each decay: where E is the excited state energy
243Am 5/2- Total angular momentum and parity need be conserved 1.1%% 10.6%% 88%% 9/2- 0.172 MeV 0.12%% 0.16%% 7/2- 0.118 MeV 5/2- 0.075 MeV 7/2+ 0.031 MeV 5/2+ 0 MeV 239Np
How fast did it happen? The mean life (often called just “the lifetime”) is defined simply as 1/ λ. That is the time required to decay to 1/e of the original population. We get:
The first Decay Rate Experiments - The Geiger –Nuttal Law As early as 1907, Rutherford and coworkers had discovered that the -particles emitted from short-lived isotopes were more penetrating (i.e. had more energy). By 1912 his coworkers Geiger and Nuttal had established the connection between particle range R and emitter half-life . It was of the form:
The one-body model of α-decay assumes that the α-particle is preformed in the nucleus, and confined to the nuclear interior by the Coulomb potential barrier. In the classical picture, if the kinetic energy of the -particle is less than the potential energy represented by the barrier height, the α-particle cannot leave the nucleus. vα Qα r=R r=b In the quantum-mechanical picture, however, there is a finite probability that the -particle will tunnel through the barrier and leave the nucleus. The α-decay constant is then a product of the frequency of collisions with the barrier, or ``knocking frequency'‘ (vα/2R), and the barrier penetration probability PT.
How high and wide the barrier? The height of the barrier is: The width of the barrier is w 30MeV Lets calculate these for taking R0=1.2F, we have