Atomic Structure and the Periodic Table
Atomic Structure and the Periodic Table. The electronic structure of an atom determines its characteristics. Studying atoms by analyzing light emissions/ absorbtions. Spectroscopy: analysis of light emitted or absorbed from a sample Instrument used = spectrometer

Atomic Structure and the Periodic Table
E N D
Presentation Transcript
The electronic structure of an atom determines its characteristics
Studying atoms by analyzing light emissions/absorbtions • Spectroscopy: analysis of light emitted or absorbed from a sample • Instrument used = spectrometer • Light passes through a slit to become a narrow beam • Beam is separated into different colors using a prism (or other device) • Individual colors are recorded as spectral lines
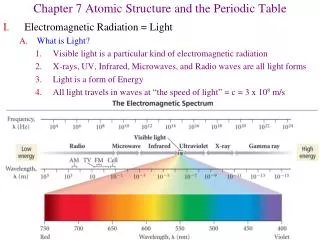
Electromagnetic radiation • Light energy • A wave of electric and magnetic fields • Speed = 3.0 x 108 m/s • Wavelength () = distance between adjacent peaks • Unit = any length unit • Frequency () = number of cycles per second • Unit = hertz (Hz)
Relationship between properties of EM waves • Wavelength x frequency = speed of light ·v = c Calculate the frequency of light that has a wavelength of 6.0 x 107 m. Calculate the wavelength of light that has a frequency of 3.7 x 1014 s-1
Visible Light • Wavelengths from 700 nm (red) to 400 nm (violet) • No other wavelengths are visible to humans
Quanta and Photons • Quanta: discrete amounts • Energy is quantized – restricted to discrete values • Only quantum mechanics can explain electron behavior • Analogy: Water flow
Another analogy for quanta • A person walking up steps – his potential energy increases in a quantized manner
Photons • Packets of electromagnetic energy • Travel in waves • Brighter light = more photons passing a point per second • Higher energy photons have a higher frequency of radiation • Planck constant h = 6.63 x 10-34Js E = hv The energy of a photon is directly proportional to its frequency
Deriving Planck’s constant In a laboratory, the energy of a photon of blue light with a frequency of 6.4 x 1014 Hz was measured to have an energy of 4.2 x 1019 J. Use Planck’s constant to show this: E = (6.63 x 10-34 J·s) x (6.4 x 10141/s) = 4.2 x 1019 J
Evidence for photons • Photoelectric effect – the ejection of electrons from a metal when exposed to EM radiation • Each substance has its own “threshold” frequency of light needed to eject electrons
Determining the energy of a photon E = hv • Use Planck’s constant! • What is the energy of a photon of radiation with a frequency of 5.2 x 1014 waves per second?
Another problem involving photon energy • What is the energy of a photon of radiation with a wavelength of 486 nm?
Louis de Broglie – proposed that matter and radiation have properties of both waves and particles (Nobel Prize 1929) • Calculate the wavelength of a hydrogen atom moving at 7.00 x 102 cm/sec • = h • m m = mass h = Planck’s constant = velocity
Hydrogen spectral lines Balmer series: n1 = 2 and n2 = 3, 4, … Lyman series (UV lines): n1= 1 and n2 = 2, 3, …
Atomic Spectra and Energy Levels • Observe the hydrogen gas tube, use the prism to see the frequencies of EM radiation emitted • Johann Balmer– noticed that the lines in the visible region of hydrogen’s spectrum fit this expression: v= (3.29 x 1015 Hz) x 1 - 1 4 n2 n = 3, 4, …
Rydberg equation: works for all lines in hydrogen’s spectrum v= RH x 1 - 1 n12 n22 RH = 3.29 x 1015 s-1 Rydberg Constant
Energy associated with electrons in each principal energy level • Energy of an electron in a hydrogen atom -2.178 x 10-18 joule E = n2 n= principal quantum number
Differences in Energy Levels of the hydrogen atom Use the Rydberg Equation OR Use the expression for each energy level’s energy in the following equation: E = Efinal – Einitial
Niels Bohr’s contribution Assumed e- move in circular orbits about the nucleus Only certain orbits of definite energies are permitted An electron in a specific orbit has a specific energy that keeps it from spiraling into the nucleus Energy is emitted or absorbed ONLY as the electron changes from one energy level to another – this energy is emitted or absorbed as a photon
Summary of spectral lines When an e- makes a transition from one energy level to another, the difference in energy is carried away by a photon Different excited hydrogen atoms undergo different energy transitions and contribute to different spectral lines
The Uncertainty Principle – Werner Heisenberg • The dual nature of matter limits how precisely we can simultaneously measure location and momentum of small particles • It is IMPOSSIBLE to know both the location and momentum at the same time
Atomic Orbitals – more than just principal energy levels • Erwin Schrodinger (Austrian) • Calculated the shape of the wave associated with any particle • Schrodinger equation – found mathematical expressions for the shapes of the waves, called wavefunctions(psi)
Born’s contribution • Max Born (German) • The probability of finding the electron in space is proportional to 2 Called the “probability density” or “electron density”
Atomic Orbital – the wavefunction for an electron in an atom • s – high probability of e- being near or at nucleus ELECTRON IS NEVER AT THE NUCLEUS IN THE FOLLOWING ORBITALS: • p – 2 lobes separated by a nodal plane • d – clover shaped • f – flower shaped
More about orbitals • Each orbital can hold 2 electrons • Orbitals in the same subshell have equal energies
Quantum numbers – like an “address” for an electron n = principal quantum number As n increases * orbitals become larger • electron is • farther from nucleus more often • higher in energy • less tightly bound to nucleus
Quantum numbers • l = angular momentum quantum number • Values: 0 to n – 1 • Defines the shape of the orbital
Quantum numbers Example: for d orbitals, m can be -2, -1, 0, 1, or 2 For p orbitals, m can be -1, 0, or 1 • ml=the magnetic quantum number • Orientation of orbital in space (i.e. pxpy or pz) • Values: between – l and l, including 0
Quantum numbers • ms = the spin number • When looking at line spectra, scientists noticed that each line was really a closely-spaced pair of lines! • Why? Each electron has a SPIN – it behaves as if it were a tiny sphere spinning upon its own axis • Spin can be + ½ or -1/2 • Each represents the direction of the magnetic field the electron creates
Describe the electron that has the following quantum numbers: Principal level 4 4p orbital px orbital spin up n = 4, l = 1, ml = -1, ms = +1/2
Are these sets of quantum numbers valid? • 3, 2, 0, -1/2 • 2, 2, 0, 1/2 NO! Level 2 2d orbital – does not exist! YES! Level 3 3d orbital 3dxz Spin down
Electron configuration: rules • Aufbau principle – electrons fill lowest energy levels first • Pauli exclusion principle – only 2 electrons may occupy each orbital, must have opposite spins • Hund’s rule – the lowest energy is attained when the number of electrons with the same spin is maximized (because electrons repel each other)
Energy level specifics 4s • s and d orbitals are close in energy • Example • 4s electrons have slightly lower energy than 3d electrons • The s electrons can penetrate to get closer to the nucleus, giving them slightly lower energy 3d
Noble Gas Configuration • A shorter electron configuration • Write the symbol for the noble gas BEFORE the element in brackets • Write the remainder of the configuration • Examples: • Cl • Cs
Special rules • One electron can move from an s orbital to the d orbital that is closest in energy • Only happens to create half or whole-filled d orbitals • Examples: Cr, Cu